ARTÍCULO ORIGINAL
doi: http://dx.doi.org/10.7705/biomedica.v35i3.2371
1 Departamento de Cirugía, Universidad de Antioquia, Medellín, Colombia; Departamento de Cirugía, Universidad de La Sabana, Chía, Colombia; Unidad de Oncología, Hospital Pablo Tobón Uribe, Medellín, Colombia
2 Departamento de Cirugía, Universidad Surcolombiana, Neiva, Colombia
Author´s contributions:
Both authors participated in posing the research question, and in the design, analysis, interpretation, and writing of the manuscript.
Recibido: 07/05/14; aceptado: 20/04/15Introduction: Thyroidectomy is a common surgery. Routine searching of the recurrent laryngeal nerve is the most important strategy to avoid palsy. Neuromonitoring has been recommended to decrease recurrent laryngeal nerve palsy.
Objective: To assess if neuromonitoring of recurrent laryngeal nerve during thyroidectomy is cost-effective in a developing country.
Materials and methods: We designed a decision analysis to assess the cost-effectiveness of recurrent laryngeal nerve neuromonitoring. For probabilities, we used data from a meta-analysis. Utility was measured using preference values. We considered direct costs. We conducted a deterministic and a probabilistic analysis.
Results: We did not find differences in utility between arms. The frequency of recurrent laryngeal nerve injury was 1% in the neuromonitor group and 1.6% for the standard group. Thyroidectomy without monitoring was the less expensive alternative. The incremental cost-effectiveness ratio was COP$ 9,112,065.
Conclusion: Routine neuromonitoring in total thyroidectomy with low risk of recurrent laryngeal nerve injury is neither cost-useful nor cost-effective in the Colombian health system.
Key words: Thyroid gland, thyroidectomy, cost-benefit analysis, meta-analysis.
doi: http://dx.doi.org/10.7705/biomedica.v35i3.2371
Análisis económico de la monitorización neurológica rutinaria del nervio laríngeo recurrente en la tiroidectomía total
Introducción. La tiroidectomía es una cirugía común. La búsqueda rutinaria del nervio laríngeo inferior es la estrategia más importante para evitar la parálisis.
Objetivo. Evaluar el costo-efectividad en un país en desarrollo de la monitorización neurológica del nervio laríngeo inferior durante la tiroidectomía.
Materiales y métodos. Se diseñó un análisis de decisiones para evaluar el costo-efectividad de la monitorización neurológica del nervio laríngeo inferior. Para las probabilidades se usaron datos de un meta-análisis. La utilidad se determinó con medidas de preferencia. Se incluyeron los costos directos. Se hizo un análisis determinístico y probabilístico.
Resultados. No se encontraron diferencias en la utilidad entre las estrategias. La frecuencia de la lesión de este nervio fue de 1 % en el grupo bajo monitorización neurológica y de 1,6 % en el grupo de control. La tiroidectomía sin monitorización fue la alternativa menos costosa. La razón de costo-efectividad incremental fue de COP$ 9.112.065
Conclusión. La monitorización neurológica rutinaria en la tiroidectomía total con bajo riesgo de lesión del nervio laríngeo inferior, no es útil con relación a su costo ni costo-efectiva en el sistema de salud colombiano.
Palabras clave: glándula tiroides, tiroidectomía, análisis costo-beneficio, metanálisis.
doi: http://dx.doi.org/10.7705/biomedica.v35i3.2371
Thyroidectomy is the most common endocrine surgery worldwide. Improvements in surgical tech nique have decreased the risk of injury to the laryngeal nerves and parathyroid glands (1,2). One of the most important complications is recurrent laryngeal nerve (RLN) injury. Most injuries are unilateral, temporary and improve in a few weeks after surgery. However, some injuries are bilateral and produce dysphonia or acute airway obstruction , thus requiring a tracheostomy, with a consequent loss in quality of life. The frequency of RLN injury, which is 1% for most thyroidectomies, can reach 12% in reoperations (1-3).
Surgical exploration with routine searching of the RLN is the most important strategy to avoid palsy. Recently, neuromonitoring has been recommended as a novel alternative to further decrease the rate of RLN palsy (4). Two published meta-analyses did not demonstrate a statistically significant difference in the rate of RLN injury (5,6). Nonetheless, some authors suggest that its use can be cost-effective based on the decrease of sequels (7), but this argument has not been tested in the literature or the Colombian health system. The aim of this study was to assess if routine neuromonitoring of RLN during total thyroidectomy is cost-effective in the Colombian health system.
Materials and methods
This is a study without risk for patients, so informed consent was not necessary. Data confidentiality was maintained by assigning a code to each patient. We designed a decision analysis study to assess the cost-effectiveness and cost-utility of routine RLN neuromonitoring during total thyroidectomy.
The target population was patients who under- went total thyroidectomy for benign and malignant disease. We excluded patients who underwent a reoperation or who had previous neck surgery; patients with a preoperative RLN injury, as demonstrated by clinical dysphonia or direct laryngoscopy or a giant goiter; or patients in which there was preoperative suspicion of local invasion of the trachea or neck vessels, because these patients were not included in published randomized controlled trials.
The strategies to be compared were neuro-moni toring in addition to routine searching of the RLN versus routine searching of RLN according to the guidelines proposed by the International Neural Monitoring Study Group (4). These guidelines include the use of a tracheal tube with an embedded electrode that acts as a receiver, an electric stimulator and an electromyography monitor that records effector muscle activity under stimulation. The stimulator, which is used to rapidly locate the RLN and assess the integrity of the nerve -muscle circuit from the vagus nerve before and after dissection, consists of four steps (preoperative and postoperative vagus and RLN stimulus). The procedure should be stopped if the electromyographic signal is lost during unilateral stimulation.
On the following postoperative day, an indirect laryngoscopy is conducted to confirm the electromyographic findings. If the vocal cord is normal, a second surgical procedure to complete the thyroidectomy is conducted in the next few days, during the same hospital stay. If the vocal cord is paralyzed, the conclusion of thyroid- ectomy is performed after vocal cord mobility is recovered. Neuromonitor use is recommended in all reoperations.
The decision tree was built with Treeage Inc. DATA 4.0 software. For the probabilities of complications, we used data from a meta-analysis of randomized controlled trials that compared both strategies. The utility was measured using preference values which are proxy variables for utilities, and the preference was considered the value that a surgeon will give to an outcome after surgery. This method was chosen due to the lack of information about the measurement of utilities of the selected outcomes with other validated instruments. Effectiveness was measured by the number of patients without definitive RLN injury.
We used the payer perspective to measure cost, because this device do not offer any long term effect on society values, different to those imposed in direct medical costs; thus, only direct costs were considered. Direct costs included surgeon and anesthetist fees, surgical time, length of hospital stay, operative resources, reoperation, clinical follow-up and the cost of treatment for complications. Because complications should be followed over the long term, a 5 year time horizon was selected, and an annual discount of 3% was applied to costs and outcomes. A net present value was calculated for utility and costs.
Data analysis
Continuous variables are shown as the mean and standard deviation, and discrete variables are shown as the frequency and range. We used the expected values for decision tree calculations.
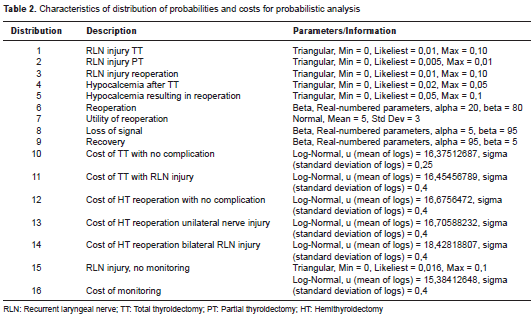
We conducted one-way and multiple-way deterministic sensitivity analyses (Tornado analysis) and a probabilistic sensitivity analysis assessing the rate of unilateral and bilateral RLN palsy, the costs of neuromonitoring and utility. One-way analysis were made changing only one variable value at a time (e.g., rate of RLN injury), while multiple-way sensitivity analysis are made changing values of all variables simultaneously (e.g. , rate of RLN injury, intraoperative loss of signal, etc.). The higher and lower values used for the sensitivity analysis were those reported in the range for each variable.
Probabilistic sensitivity analysis uses a simulated scenario where values of different variables change by hazard and the result of each trial is drawn in a cost-effectiveness plane. For the probabilistic analysis, we assumed a b and triangular distribution for categorical variables (e.g., frequency of palsy) and a normal and log normal distribution for costs, and conducted 1,000 iterations during a Monte Carlo simulation. Finally, a worst scenario analysis was conducted using cost from the literature and information from Gardner, et al . (8), in which a unilateral RLN definitive palsy increased costs by 65% compared to no palsy.
Results
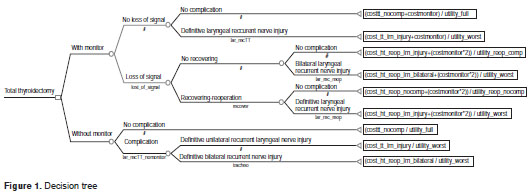
The decision tree was designed following the clinical pathway shown in figure 1.

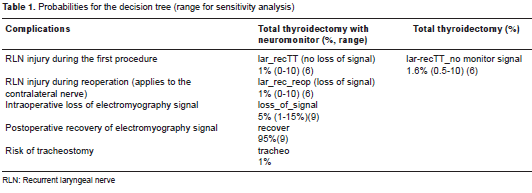
Probabilities
The probabilities of RLN injury were obtained from a previous meta-analysis (6). For loss of signal probability, we used information from a study by Sitges-Serra , et al. (9) (table 1). We assumed that the risk of each individual RLN is similar and independent of the contralateral nerve because neuromonitoring guidelines recommend stopping the surgical procedure if a loss of signal occurs during the dissection of the first nerve. According to this guideline, the risk of bilateral RLN palsy corresponds to 1% of all cases of unilateral RLN palsy. For probabilistic analysis, characteristics of each distribution are shown in table 2.
Utility
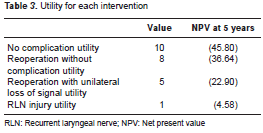
We assigned a utility value to each outcome on a scale from 1 to 10, corresponding to surgeon preference for this outcome. We conducted a survey to obtain baseline utility estimates for each of the health outcome states, asking to head and neck surgeons about the value they gave to each state, as described by Kebebew, et al . (10). The value of 10 was assigned to the scenario without any complications or reoperation. The value of 1 was assigned to the scenario with RLN definitive palsy. A value of 8 was assigned to the situation in which there are no complications but reoperation is necessary (false positive values of intraoperative neuromonitoring), and a value of 5 was assigned to situations with no recovery from unilateral palsy after neuromonitor surgery (true positives) and contralateral uncomplicated surgery (table 3). All utility values were adjusted to net present value at 5 years.
Effectiveness
Effectiveness was measured as the number of patients without nerve injury, assuming a cohort of 100 patients.
Costs
We used Colombian pesos to calculate costs (1 US dollar = 1,800 Colombian pesos, 2013). The costs were obtained using micro-costing from 50 charts of patients who underwent a total thyroidectomy by the same surgeon at a tertiary care hospital in Colombia. All resources used during the surgical procedure were collected using a previously designed form. For follow-up, we recorded the number of physician visits, medications, vocal therapy treatments and procedures to calculate an annual value by apportioning according to the months of follow-up. The final cost for each patient was calculated by multiplying each resource by its standard price, which was obtained from a national price manual (SOAT 2011) or by searching public bids available at government web sites (11). Finally, we conducted a Monte Carlo simulation with 500 iterations to consider cost variations; the mean of this simulation was considered to be the mean cost, and the upper and lower limits were the probable range of costs.
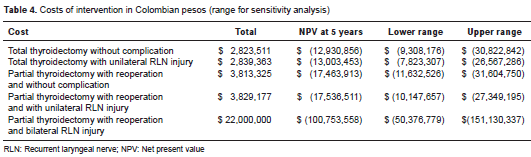
We could not find cases of bilateral RLN palsy. Therefore, we assumed that the cost in this case should include a tracheostomy, a three day ICU stay with a daily value of COP$ 1,000,000 and an additional four day hospital stay for a total value of COP$ 10,000,000. For follow-up costs, this patient most likely will require pulmonary therapy and tra cheostomy care with an additional value of COP$ 1,000,000/month. This cost represents increase of greater than 10 times compared to a patient without any complications. For the neuromonitoring arm, we used the value of renting a device and the value associated with the use of the device. We did not consider the capital cost of purchasing the equipment because renting is common in Colombia. The monitor value was calculated to be COP$ 4,800,000 (COP$ 1,500,000 for the tracheal tube and COP$ 3,300,000 for renting the monitor). According to published guidelines, the monitor value was duplicated in cases of reoperation. Table 4 shows the direct cost values.
Utility analysis
The deterministic analysis did not find differences in utility between both arms (44.9 units for the monitor vs. 45.1 units without a monitor ).
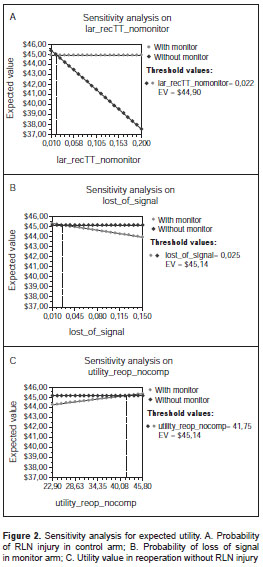
The one-way sensitivity analysis showed that the monitor arm has greater utility in cases of RLN injury, with a frequency greater than 2.2% in the control arm, when the loss of signal frequency was lower than 2.5% and the utility assigned to reoperation without complication was higher than 9 units. However, this increase was small (from 4 5.1 to 45.8 in utility units ) and not clinically significant. The rate of RLN injury in reoperation, rate of recovery after loss of signal and utility value in reoperation with complication did not have any effect on the analysis (figure 2). The tornado analysis showed that most important variables were RLN injury frequency in the control arm, utility of reoperation without complication and loss of signal frequency. Therefore, it can be assumed that the utility is clinically similar in both strategies.
Effectiveness analysis
The data from the recent meta-analysis showed that the frequency of RLN injury was 1% in the neuromonitor group and 1.6% for the standard group, which was a no significant difference (6).
Cost analysis
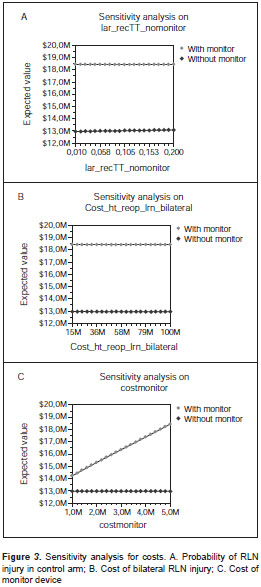
In the deterministic analysis, total thyroidectomy without a monitor was the less expensive alternative (COP$ 12,942,737 vs. COP$ 18,409,976). The one way sensitivity analysis showed that total thyroidectomy with a monitor dominated all possible scenarios (figure 3). The tornado analysis showed that most important variables were RLN injury frequency in the control arm, bilateral RLN injury cost and monitor cost. Therefore, total thyroidectomy without a monitor is assumed to be less expensive.
Cost-utility and cost-effectiveness analysis
The cost-utility and cost-effectiveness analysis results are shown in table 5. For each unit of utility, it is necessary to pay COP$ 286.979 in the non-monitor strategy compared with COP$ 410,022 in the monitor arm, with a negative incremental cost-utility ratio of COP$ 27,336,195. Each effectiveness unit (RLN injury avoided) corresponds to COP$ 131,531.88 for the non-monitor arm compared with COP$ 185,989 for the monitor arm, with an incremental cost-effectiveness ratio of COP$ 9,112,065. Therefore, total thyroidectomy without a monitor is the best strategy in the deterministic analysis .
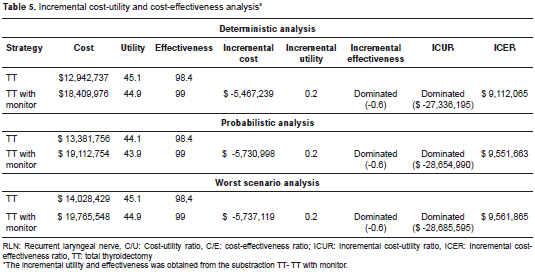
Figure 4 shows the probabilistic analysis of the mean cost-utility for both alternatives. It is clear that the incremental cost-utility graph shows that 63% of the iterations are located in quadrant II, and most iterations are located to the left of one unit of utility, representing non-clinically significant differences in utility. Figure 5 shows the acceptability curve, in which the non-monitor arm is the best option, even when COP $ 40,000,000 is the willingness to pay.
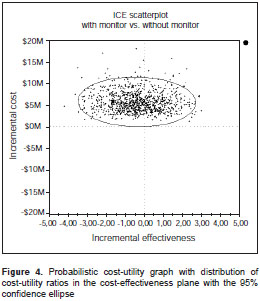
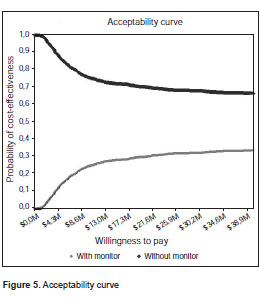
The conclusions did not change following the assessment of the worst case scenario.
Discussion
Injury of the RLN during total thyroidectomy is a very uncommon event that greatly impacts the quality of life. Throughout the history of thyroid surgery, many strategies have been designed to avoid RLN injury, and the identification and visualization of the nerve have demonstrated its utility. Specifically, this strategy has demonstrated an RLN injury rate lower than 1% in specialized centers (12). Many reports have suggested that intraoperative neuromonitoring could further decrease the risk of RLN injury during thyroid surgery (7,13-16). Nonetheless, recent meta-analyses, one of which included observational and experimental studies (5) and another that only included randomized controlled trials (6), did not find a significant difference in RLN injury frequency when comparing neuromonitoring with the standard procedure. However, some authors still recommend the use of neuromonitoring, and the costs of implementation are unknown (17,18). Because the cost structure cannot be extrapolated across health systems, we conducted this study to assess the cost-utility and cost-effectiveness of neuromonitoring in Colombia (19,20).
It was clear that both alternatives have clinically equivalent utility and effectiveness. The lack of significant differences in the RLN injury rate demon strated in recent meta-analyses and the utility analysis in this study indicate that surgeons prefer both strategies to a similar extent and demonstrate that a new, more expensive device does not offer clinical advantages in this case. The cases when utility was higher for the monitoring arm only represented an increase in 0.7 units in a scale of 1 to 10, which are clinically non-significant.
This study showed that neuromonitoring resulted in a dramatic increase in costs due to the large impact of the device on cost structure. There was an approximately 40% increase in the cost of the neuromonitoring alternative, and the lack of cost- effectiveness was maintained with modifications of clinical variables such as the RLN injury frequency and the operative characteristics of neuromonitoring (loss of signal during the procedure) in the range presented. The deterministic analysis showed that neuromonitoring was associated with an extra cost of almost 27 million Colombian pesos for each utility unit gained and nine million Colombian pesos for each patient without an RLN injury. These data were confirmed in a probabilistic model that introduces a random effect into the comparisons and showed that most cases with neuromonitoring were less cost-effective with a mean incremental cost- utility ratio of approximately nine million Colombian pesos for each utility unit gained. The poor cost-effectiveness of the use of this device can be understood by comparing its costs with the value of the total thyroidectomy procedure, including a one day stay in the Colombian health system, which costs around three million Colombian pesos.
However, it is important to recognize some weak-nesses that can affect the study conclusions. We did not include surgical time in the calculation of intraoperative costs. Some authors have shown that neuromonitoring can decrease operative time. Nonetheless, the decrease in operative time is around seven minutes, and we believe that this small change does not affect the final conclusions (21). Additionally, we only included direct costs and payer perspective in the analysis. It is possible that a consideration of the societal perspective and inclusion of indirect costs can modify the overall values, but we believe that the final conclusion will not change in the absence of differences in effectiveness and utility. It is important to note that the target population used in this analysis included only total thyroidectomy patients with a low risk of RLN injury, such as patients with small thyroid glands and without previous neck surgery or local invasion. The results of other specific cases could be different. Besides, the method to elicit utilities was made with a preference scale. It has been shown that other methods to measure utilities, as validated specific scales, have a better performance than analog scales and that utilities measured by physicians and normal patients undervalue effects on patients. However, the lack of information about measurements with specific QOL instruments in this type of patients impeded its use in the model. Therefore, we believe that utilities values used in this model could be even lower and that results of cost-utility ratios could be even greater than those calculated here (22,23). Finally, costs were calculated using the SOAT manual, that could overestimate them over other costs sources as the ISS manual, but we believe conclusions will not change because the greater cost comes from a device not included in any manual, with similar values for both costs calculation systems.
Routine neuromonitoring in total thyroidectomy with a low risk of RLN injury is neither cost-useful nor cost-effective in the Colombian health system, and its use introduces an important burden to the cost structure of a very common surgical procedure. Given that the epidemic of thyroid cancers, most of which are asymptomatic, smaller than 2 cm and found in young people, corresponds to a low risk of nerve injury, the use of routine neuromonitoring should not be recommended for most patients in Colombia and this results can be useful to other developing countries. The cost-effectiveness of neuromonitoring in cases with a high risk of nerve injury should be evaluated before making any recommendation.
Authors were invited to a training meeting by the distributor of the device.
Authors did not receive any external funding.
Corresponding author: Álvaro Sanabria, Unidad de Oncología, Hospital Pablo Tobón Uribe, Carrera 51D N° 62-29, Medellín, Colombia Teléfono: (574) 219 6000 alvarosanabria@gmail.com
1. Zambudio AR, Rodríguez J, Riquelme J, Soria T, Canteras M, Parrilla P. Prospective study of postoperative complications after total thyroidectomy for multinodular goiters by surgeons with experience in endocrine surgery. Ann Surg. 2004;240:18-25. http://dx.doi.org/00000658-200407000-00004
2. Karamanakos SN, Markou KB, Panagopoulos K, Karavias D, Vagianos CE, Scopa CD, et al . Complications and risk factors related to the extent of surgery in thyroidectomy. Results from 2,043 procedures. Hormones (Athens). 2010;9:318-25.
3. Filho JG, Kowalski LP. Postoperative complications of thyroidectomy for differentiated thyroid carcinoma. Am J Otolaryngol. 2004;25:225-30 . http://dx.doi.org/ S0196070904000262
4. Randolph GW, Dralle H, Abdullah H, Barczynski M, Bellantone R, Brauckhoff M, et al . Electrophysiologic recurrent laryngeal nerve monitoring during thyroid and parathyroid surgery: International standards guideline statement. Laryngoscope. 2011;121(Suppl.1):S1-16. http://dx.doi.org/10.1002/lary.21119
5. Higgins TS, Gupta R, Ketcham AS, Sataloff RT, Wadsworth JT, Sinacori JT. Recurrent laryngeal nerve monitoring versus identification alone on post-thyroidectomy true vocal fold palsy: A meta-analysis. Laryngoscope. 2011;121:1009-17. http://dx.doi.org/10.1002/lary.21578
6. Sanabria A, Ramírez A, Kowalski L, Silver C, Shaha A, Owen R, et al . Neuromonitoring in thyroidectomy: A meta-analysis of effectiveness from randomized controlled trials. Eur Arch Otorhinolaryngol. 2013;270:2175-89. http://dx.doi.org/10.1007/s00405-013-2557-2
7. Dueñas JP, Duque CS. Monitorización intraoperatoria de los nervios laríngeos superior e inferior en cirugía de tiroides y paratiroides. Rev Colomb Cir. 2012;27:298-305.
8. Gardner GM, Smith MM, Yaremchuk KL, Peterson EL. The cost of vocal fold paralysis after thyroidectomy. Laryngoscope. 2013;123:1455-63. http://dx.doi.org/10.1002/lary.23548
9. Sitges-Serra A, Fontane J, Dueñas JP, Duque CS, Lorente L, Trillo L, et al . Prospective study on loss of signal on the first side during neuromonitoring of the recurrent laryngeal nerve in total thyroidectomy. Br J Surg. 2013;100:662-6. http://dx.doi.org/10.1002/bjs.9044
10. Kebebew E, Duh QY, Clark OH. Total thyroidectomy or thyroid lobectomy in patients with low-risk differentiated thyroid cancer: Surgical decision analysis of a controversy using a mathematical model. World J Surg. 2000;24:1295-302. http://dx.doi.org/10.1007/s002680010215
11. Sanabria A, Gómez X, Domínguez L, Vega V, Osorio C. Tiroidectomía total basada en la evidencia, análisis de impacto presupuestario. Rev Colomb Cir. 2012;27:30-9.
12. Lahey FH, Hoover WB. Injuries to the recurrent laryngeal nerve in thyroid operations: Their management and avoidance. Ann Surg. 1938;108:545-62.
13. Barczynski M, Konturek A, Cichon S. Randomized clinical trial of visualization versus neuromonitoring of recurrent laryngeal nerves during thyroidectomy. Br J Surg. 2009;96:240-6. http://dx.doi.org/10.1002/bjs.6417
14. Dionigi G, Alesina PF, Barczynski M, Boni L, Chiang FY, Kim HY, et al . Recurrent laryngeal nerve injury in video-assisted thyroidectomy: Lessons learned from neuro- monitoring. Surg Endosc. 2012;26:2601-8. http://dx.doi.org/10.1007/s00464-012-2239-y
15. Durán MC, Dionigi G, Sitges-Serra A, Barczynski M, Angelos P, Dralle H, et al . Intraoperative monitoring of the recurrent laryngeal nerve during thyroidectomy: A standardized approach part 2. World Journal of Endocrine Surgery. 2012;4:33-40. http://dx.doi.org/10.5005/jp-journals- 10002-1091
16. Durán MC, Dionigi G, Sitges-Serra A, Barczynski M, Angelos P, Dralle H, et al . Intraoperative monitoring of the recurrent laryngeal nerve during thyroidectomy: A standardized approach (Part 1). World Journal of Endocrine Surgery. 2011;3:144-50. http://dx.doi.org/10.5005/jp-journals-10002-1079
17. Beccagutti G, Grifi M, Pantaleoni M, Dionigi G. The impact of neuromonitoring on thyroid surgery costs. Value in Health. 2010;13:A289. http://dx.doi.org/10.1016/S1098-3015(11)72089-7
18. Dionigi G, Bacuzzi A, Boni L, Rausei S, Rovera F, Dionigi R. Visualization versus neuromonitoring of recurrent laryngeal nerves during thyroidectomy: What about the costs? World J Surg. 2012;36:748-54. http://dx.doi.org/10.1007/s00268-012-1452-0
19. Bryan S, Brown J. Extrapolation of cost-effectiveness information to local settings. J Health Serv Res Policy. 1998;3:108-12.
20. Drummond MF, Bloom BS, Carrin G, Hillman AL, Hutchings HC, Knill-Jones RP, et al . Issues in the cross-national assessment of health technology. Int J Technol Assess Health Care. 1992;8:671-82. http://dx.doi.org/10.1017/S0266462300002361
21. Dionigi G, Boni L, Rovera F, Bacuzzi A, Dionigi R. Neuromonitoring and video-assisted thyroidectomy: A prospective, randomized case-control evaluation. Surg Endosc. 2009;23:996-1003. http://dx.doi.org/10.1007/s00464-008-0098-3
22. Sneeuw KC, Aaronson NK, Sprangers MA, Detmar SB, Wever LD, Schornagel JH. Evaluating the quality of life of cancer patients: Assessments by patients, significant others, physicians and nurses. Br J Cancer. 1999;81:87-94 http://dx.doi.org/10.1038/sj.bjc.6690655
23. Blazeby JM, Williams MH, Alderson D, Farndon JR. Observer variation in assessment of quality of life in patients with oesophageal cancer. Br J Surg. 1995;82:1200-3. http://dx.doi.org/10.1002/bjs.1800820916