COMUNICACIÓN BREVE
Grupo de Microbiología, Instituto Nacional de Salud, Bogotá, D.C., Colombia
Author´s contributions:
Carolina Duarte and Jaime Moreno conceived and designed the experiments.
Olga Sanabria, Sandra Yamile Saavedra and Karina Rodríguez performed the experiments.
Carolina Duarte, Jaime Moreno and Sandra Saavedra analyzed the data.
Jaime Moreno wrote the paper.
Recibido: 11/09/14; aceptado: 13/12/14
Introduction: Neisseria meningitidis is one of the leading causes of bacterial meningitis. In 2013, an increase in invasive disease caused by N. meningitidis serogroup B occurred in Cartagena, Colombia.
Objective: The aim of this study was to characterize serogroup B isolates recovered from patients with meningococcal meningitis in Cartagena during 2012 and 2014.
Materials and methods: Twenty-five isolates were collected through a national, laboratory-based surveillance, and phenotypic and genotypic characteristics were determined by serosubtyping, serotyping, antimicrobial susceptibility testing, repetitive sequence-based PCR (rep-PCR). MLST data available were also used.
Results: The B:10,15:nt phenotype, associated with clonal complex ST-41/44, was responsible for 68.0% of all cases.
Conclusion: The clinical, epidemiological and molecular characterization of meningococcal disease is necessary for understanding the spread of virulent meningococcal clones and for facilitating the early detection of cases to reduce epidemic risks.
Key words: Neisseria meningitidis , serogroup B; meningitis, phenotype.
doi: http://dx.doi.org/10.7705/biomedica.v35i1.2414
Caracterización fenotípica y genotípica de aislamientos de Neisseria meningitidis, serogrupo B, procedentes de Cartagena, Colombia, 2012-2014
Introducción. Neisseria meningitidis es una de las causas principales de meningitis bacteriana. En el 2013 se evidenció un aumento de la enfermedad invasiva causada por N. meningitidis, serogrupo B, en Cartagena, Colombia.
Objetivo. El objetivo de este estudio fue caracterizar los aislamientos de N. meningitidis, serogrupo B, recuperados de pacientes con meningitis meningocócica en Cartagena durante el año 2013.
Materiales y métodos. Se recolectaron 25 aislamientos a través de la vigilancia basada en el laboratorio nacional y se determinaron las características fenotípicas y genotípicas mediante serosubtipificación, serotipificación, pruebas de sensibilidad a los antimicrobianos, amplificación de secuencias de ADN repetidas (REP-PCR) y el uso de datos disponibles en el sitio de MLST.
Resultados. El fenotipo B:10,15:nt, asociado con el complejo clonal ST-41/44, fue responsable del 68,0 % de todos los casos.
Conclusión. La caracterización clínica, epidemiológica y molecular de los aislamientos causantes de enfermedad meningocócica es necesaria para el conocimiento de la propagación de clones virulentos y facilita la detección temprana de casos, lo que reduce los riesgos de epidemia.
Palabras clave: Neisseria meningitidis , serogrupo B; meningitis, fenotipo.
doi: http://dx.doi.org/10.7705/biomedica.v35i1.2414
Neisseria meningitidis has become the leading cause of bacterial meningitis in children, especially after the introduction of Streptococcus pneumoniae and Haemophilus influenzae type b routine vaccination (1). There are 12 serogroups of N. meningitidis ; however, the majority of invasive meningococcal infections is caused by serogroups A, B, C, W, X and Y, and the distribution varies with the geographical location and the age group (2). The overall incidence of meningococcal disease differs from one region to another ranging between <2 cases/100,000 population per year in the United States, China and European and Latin American countries, and >10/100,000 population per year in some African countries (3,4).
N. meningitidis serogroup B has emerged as an important cause of invasive meningococcal disease in some regions of Europe, Latin America and North America since the introduction of vaccines against serogroups A and C (5). Despite the genetic heterogeneity of serogroup B strains, two hypervirulent clonal complexes, ST-41/44 3 and ST-32 , are responsible for the majority of serogroup B disease cases in several countries (5). In 2012, 33 cases of meningococcal disease were reported in Colombia, of which 69.7% were caused by N. meningitidis serogroup B (6). In 2012, an increase in invasive disease caused by N. meningitidis serogroup B (25 cases in total) was observed in Cartagena de Indias, Colombia, which has been declared by UNESCO as a world heritage site. In the present study, we characterized N. meningitidis serogroup B isolates recovered between 2012 and 2014 from infected patients living in deprived urban areas in Cartagena.
Materials and methods
We analyzed 25 N. meningitidis isolates recovered between March, 2012, and October, 2014, from cerebrospinal fluid (CSF) or blood samples of meningococcal disease patients. The isolates were received at the Colombian Instituto Nacional de Salud through the SIREVA II international surveillance network (6). We confirmed isolates by using standard microbiology techniques, while the serogroup was determined by slide agglutination with commercial antiserum against meningo- coccal capsular polysaccharides (DIFCO, Beckton Dickinson). Serosubtyping and serotyping were done by dot blot with monoclonal antibodies (RIVM, Bilthoven, the Netherlands, and Institute Adolfo Lutz, São Paulo, Brazil) (7). Agar dilution antimicrobial susceptibility testing for penicillin (P), ceftriaxone (CRO), chloramphenicol (CL), ciprofloxacin (CIP) and rifampin (RIF) was performed according to the Clinical and Laboratory Standards Institute guidelines (8).
We performed genotyping on the 19 isolates using the bacterial rep-PCR-DiversiLab kit (Bacterial BarCodes, bioMérieux Inc., Houston, TX) following the manufacturer´s recommendations. We analyzed patterns by using the Pearson correlation coefficient to create a dendrogram of the genetic relationship among isolates (9). Clusters of rep-PCR patterns exhibiting a similarity of >93% were designated by capital letters. Additionally, we included data from the Multi Locus Sequence Typing (MLST) site for four isolates recovered in 2010 (isolate 858), 2011 (isolates 903 and 905) and 2012 (isolate 926), which were studied as part of a SIREVA II Latin American project. MLST was performed according to the methods of Maiden, et al . Primers, as well as the determination of sequence alleles and the designation of sequence types, were retrieved from the MLST website ( http://neisseria.org/nm/typing/mlst ).
Results
The 25 N. meningitidis serogroup B isolates were sent by six hospital laboratories, but 50% of them were recovered in only one hospital. All patients came from a geographically defined area in 12 of the 250 city districts characterized by high levels of poverty and low educational levels.
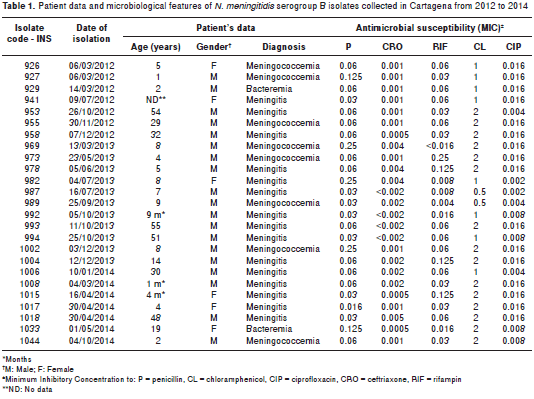
Seventy six per cent of meningococcal disease was diagnosed in men; patients´ ages ranged from one month to 55 years: 8 isolates (32.0%) were recovered from patients <5 years of age; 8 (32.0%), from patients 5 to 14 years of age, and 8 (32.0%) from patients >14 years of age; no information was available for one of the patients (4.0%). Isolates were recovered from blood (60.0%) and CSF (40.0%) samples. Patients had been diagnosed with meningitis (60.0%), meningococcemia (32.0%) and bacteremia (8.0%). Two (8.0%) were penicillin- non-susceptible isolates (0.250 µg/mL), whereas all of them were susceptible to CRO, CL, CIP and RIF (table 1).

The most prevalent serotype and serosubtype was B:10,15:nt (60.0%), followed by B:1:P1.22-1,14 (16.0%) (figure 1). Isolates were grouped in three rec-PCR clusters (A, B, C) designated by size, of which A was the largest (figure 1). Cluster A, grouping 17 isolates from the study and 2 controls (isolates 903 and 905), was B:10,15:nt and showed genetic relation with the clonal complex ST-41/44. Three B:1:P1.22-1,14 isolates were grouped as cluster B, and five isolates with different serotype and serosubtype were grouped in cluster C. Isolate 858 from 2010 was B:4,7:P1.1 and the clonal complex was ST23.

Discussion
Using molecular techniques, we characterized invasive N. meningitidis serogroup B isolates that had circulated in Cartagena as part of a meningococcal disease epidemic affecting low socioeconomic population groups considered at high risk for the disease (10). Serogroup B has become a recent threat with sporadic, endemic and epidemic occurrences being recorded in North America, Europe, South America and Australia (5). From 1987 through 2012, 680 N. meningitidis isolates were confirmed in Colombia by the Microbiology Group of the Instituto Nacional de Salud, of which 72.2% (n=491) were serogroup B (6).
Overall, isolates were susceptible to most of the antibiotics tested. SIREVA II data showed that between 2006 and 2010, N. meningitidis isolates recovered in Bolivia, Colombia, Ecuador, Perú and Venezuela corresponded predominantly to sero- groups B and Y (41.7% and 29.1%, respectively). Decreased sensitivity to P was found in 17.1% of isolates (16.1% intermediate and 1.0% fully resis tant isolates) while all of them remained highly susceptible to RIF (99.5%) (11). A systematic review of the effectiveness of different antibiotics for prophylaxis against meningococcal disease found that penicillin and rifampin proved effective for eradicating N. meningitidis ; however, the use of rifampin may lead to the emergence of resistant strains (12). Therefore, antimicrobial chemopro phylaxis for close contacts of patients with invasive meningococcal disease could be important to prevent further cases (12).
The information on the distribution of N. meningitidis serogroup B isolates serosubtypes and serotypes is important in evaluating the possibility of using outer membrane protein (OMP) based vaccines to control endemic meningococcal disease (13). However, the variability in OMP strains causing endemic disease will likely limit their usefulness (10). The main serosubtype and serotype (B:10,15:nt) identified in this study differ from those in the vaccines currently licensed: The Cuban VA-MENGOC-BC vaccine, which was developed from the outer membrane antigens of the epidemic strain type B:4:P1.15 combined with serogroup C polysaccharide (14 ), and a multicomponent serogroup B meningococcal vaccine (4CMenB), consisting of factor-H binding protein (fHbp), N. meningitidis adhesin A (NadA) and Neisseria heparin binding antigen (NHBA), combined with outer membrane vesicles from the New Zealand epidemic strain NZ98/254 (B:4:P1.7b,4) (15). This vaccine was approved for use in the European Union and Australia, and has been found to be safe and immunogenic in a Latin American adolescent population (10,16). The predicted protection of this new vaccine is dependent of vaccine antigens expressed by strains causing disease in different geographical settings, therefore, fHbp, NadA and NHBA genetic typing of invasive N. meningitidis serogroup B isolates is required.
A limited number of clonal complexes are responsible for the majority of the disease cases associated with outbreaks or epidemics. In our study, the most common clonal complex was ST-41/44, found in 64.3% of cases. The ST-41/44 is the most diverse clonal complex associated with serogroup B meningococcal disease, and the analysis of available MLST data showed that the most common serotypes in this complex are 41, 44, 42, 40 and 154 (5). This clonal complex has caused epidemics in the Netherlands and New Zealand and was the dominant cause of invasive meningococcal disease in Ireland, Belgium and Italy (5). In Chile, the ST-41/44 complex, mostly associated with B:P1.19,13-1, showed the highest prevalence (29.0%), (17), while in Argentina, only 7% of the serogroup B isolates in 2010 belonged to the ST-41/44 complex (18).
Phenotyping and genotyping methods are impor tant for meningococcal transmission studies and epidemics early detection. Additionally, molecular surveillance is crucial to identify genotypes indicating disease common sources, as well as to determine the antigenic profile and clonal diversity of N. meningitidis isolates associated with invasive disease, and to monitor the evolution and distribution of meningococcal serogroups and clones in order to provide information for future vaccine development.
Acknowledgments
The authors thank Elizabeth Castañeda and Clara Inés Agudelo for their critical review of the manuscript and Orlando Castillo for his help in data management. We also thank the personnel of hospital microbiology laboratories in Cartagena for identifying the N. meningitidis cases and sending the isolates.
Conflicts of interest
None declared by the authors.
Funding
This work was financed by the Instituto Nacional de Salud, Bogotá, Colombia. The funders had no role in data collection and analysis, decision to publish, or preparation of the manuscript.
Corresponding author: Jaime Moreno, Grupo de Microbiología, Instituto Nacional de Salud, Avenida calle 26 No 51-20, Bogotá, D.C., Colombia Phone: (571) 220 7700, extension 1420; telfax: (571) 220 7700, extension 1421
jmoreno@ins.gov.co1. McIntyre PB, O´Brien KL, Greenwood B, van de Beek D. Effect of vaccines on bacterial meningitis worldwide. Lancet. 2012;380:1703-11. http://dx.doi.org/10.1016/S0140-6736(12)61187-8
2. Jafri RZ, Ali A, Messonnier NE, Tevi-Benissan C, Durrheim D, Eskola J, et al. Global epidemiology of invasive meningococcal disease. Popul Health Metr. 2013;11:17. http://dx.doi.org/10.1186/1478-7954-11-17
3. Read RC. Neisseria meningitidis ; clones, carriage and disease. Clin Microbiol Infect. 2014;20:391-5. http://dx.doi. org/10.1111/1469-0691.12647
4. Sáfadi MA, Cintra OA. Epidemiology of meningococcal disease in Latin America: Current situation and opportunities for prevention. Neurol Res. 2010;32:263-71. http://dx.doi.org/10.1179/016164110X12644252260754
5. Racloz VN, Luiz SJ. The elusive meningococcal meningitis serogroup : A systematic review of serogroup B epidemiology. BMC Infect Dis. 2010:10;175. http://dx.doi. org/10.1186/1471-2334-10-175
6. Sistema de Redes de Vigilancia de los Agentes Responsables de Neumonías y Meningitis Bacterianas (SIREVA II). Informe Regional de SIREVA II, 2012. 2013. Fecha de consulta: 1° de diciembre de 2013.
7. Wedege E, Hoiby EA, Rosenqvist E, Froholm O. Serotyping and subtyping of Neisseria meningitidis isolates by coagglutination, dot blotting and ELISA. J Med Microbiol. 1990:31;195-201. http://dx.doi.org/10.1099/00222615-31-3-195
8. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty- First Informational Supplement. CLSI document M100-S21. Wayne, PA: CLSI; 2013.
9. Healy M, Huong J, Bittner T, Lising M, Frye S, Raza S, et al . Microbial DNA typing by automated repetitive-sequence-based PCR. J Clin Microbiol. 2005:43;199-207. http://dx.doi.org/10.1128/JCM.43.1.199-207.2005
10. Cohn AC, MacNeil JR, Clark TA, Ortega-Sánchez IR, Briere EZ, Meissner HC, et al . Prevention and control of meningococcal disease: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2013:62;1-28.
11. Ibarz-Pavón AB, Lemos AP, Gorla MC, Regueira M; SIREVA Working Group II, Gabastou JM. Laboratory- based surveillance of Neisseria meningitidis isolates from disease cases in Latin American and Caribbean countries, SIREVA II 2006-2010. PLoS One. 2012:7;e44102. http:// dx.doi.org/10.1371/journal.pone.0044102
12. Zalmanovici Trestioreanu A, Fraser A, Gafter-Gvili A, Paul M, Leibovici L. Antibiotics for preventing menin-gococcal infections. Cochrane Database Syst Rev. 2013; 10:CD004785. http://dx.doi.org/10.1002/14651858.CD004785
13. Tondella ML, Popovic T, Rosenstein NE, Lake DB, Carlone GM, Mayer LW, et al. Distribution of Neisseria meningitidis serogroup B serosubtypes and serotypes circulating in the United States. J Clin Microbiol. 2000:38:3323-8.
14. González de Aledo A, Viloria L. Meningococcus B serosubtypes causing invasive disease in Cantabria [Spain] and agreement with the Cuban vaccine strain. Gac Sanit. 2004.18:45-9.
15. O´Ryan M, Stoddard J, Toneatto D, Wassil J, Dull PM. A multi-component meningococcal serogroup B vaccine (4CMenB): The clinical development program. Drugs. 2014:74:15-30. http://dx.doi.org/10.1007/s40265-013-0155-7
16. Rüttimann RW, Gentile A, Parra MM, Saez-Llorens X, Sáfadi MA, Santolaya ME. A consensus statement: Meningococcal disease among infants, children and adolescents in Latin America. Pediatr Infect Dis J. 2014.33:284- 90. http://dx.doi.org/10.1097/INF.0000000000000228
17. Barra GN, Araya PA, Fernández JO, Gabastou JM, Hormazábal JC, Seoane M, et al . Molecular characterization of invasive Neisseria meningitidis strains isolated in Chile during 2010–2011. PLoS ONE. 2013.8: e66006. http://dx.doi.org/10.1371/journal.pone.0066006
18. Sorhouet-Pereira C, Efron A, Gagetti P, Faccone D, Regueira M, Corso A, et al . Phenotypic and genotypic characteristics of Neisseria meningitidis disease causing strains in Argentina, 2010. PLoS ONE. 2013.8:e58065. http://dx.doi.org/10.1371/journal.pone.0058065