ARTÍCULO ORIGINAL
doi: http://dx.doi.org/10.7705/biomedica.v33i2.694
Yurika López1, Francisco Vargas2, Mónica Velásquez2, Carolina Ortiz1, Libia M. Rodríguez1, Sara París1, Luis F. García1, Andrés Baena1, Gloria Vásquez1, 2
1 Grupo de Inmunología Celular e Inmunogenética, Facultad de Medicina, Universidad de Antioquia, Medellín, Colombia
2 Grupo de Reumatología, Facultad de Medicina, Universidad de Antioquia, Medellín, Colombia
Author contributions: Yurika López performed the experiments, collected and analyzed the data, and wrote the paper. Francisco Vargas and Mónica Velásquez recruited and evaluated the patients. Carolina Ortiz performed the experiments and collected data. Libia M. Rodríguez collected and analyzed the data and wrote and critically reviewed the manuscript. Sara París supervised the experiments. Luis F. García analyzed the data and critically reviewed the manuscript. Andrés Baena critically reviewed the manuscript. Gloria Vásquez recruited and evaluated the patients, conceived and designed the experiments, collected and analyzed the data, and wrote the paper
Recibido: 29/03/12; aceptado:23/11/11Introduction. Rheumatoid arthritis patients under treatment with anti-TNF-α are at a high risk of developing active tuberculosis, and therefore, screening for latent tuberculosis infection is recommended before anti-TNF-α therapy.
Objective. To compare the tuberculin test and IFNγ production induced by culture filtrate proteins(CFPs) and Mycobacterium tuberculosis-specific CFP-10 antigens in rheumatoid arthritis patients.
Materials and methods. An analytic transversal study was conducted in rheumatoid arthritis patients treated at Hospital Universitario San Vicente Fundación between January and December 2007. IFNγ production in response to CFPs and CFP-10 was measured in the supernatants of whole blood cultures and evaluated for correlations with tuberculin reactivity. The degree of concordance between both tests was also established.
Results. Forty-five patients were included, of which 14 (31.1%) had a tuberculin reaction of ≥10 mm of induration, 9 (20%) produced IFNγ in response to CFP-10, and 7 were positive for both tests. The correlation between tests was r=0.53 (IC 95%:0.28-0.72), and the global concordance between tests was80%, with a Kappa coefficient of 0.48 (IC95%:0.20-0.76).
Conclusions. Only two tuberculin (-)/CFP-10+ “anergic” patients were observed. By contrast, six tuberculin +/CFP-10(-) “tuberculin false-positive” patients were observed. These data suggest that the tuberculin test is not an appropriate tool for determining the need for tuberculosis prophylaxis.
Keywords: Arthritis, rheumatoid; latent tuberculosis; tuberculin test; interferon gamma release assay; interferon-gamma; tumor necrosis factor-alpha/antagonists and inhibitors.
doi: http://dx.doi.org/10.7705/biomedica.v33i2.694
Correlación de la respuesta a antígenos de Mycobacterium tuberculosis y la prueba cutánea de tuberculina en pacientes con artritis reumatoide
Introducción. Los pacientes con artritis reumatoide bajo tratamiento con anti-TNFα están en alto riesgo de desarrollar tuberculosis activa, por lo cual se recomienda hacer la tamización para infección latente de tuberculosis, antes de iniciar el tratamiento.
Objetivo. Comparar la prueba de tuberculina y la producción de IFNγ inducida por antígenos CFP (Culture Filtrate Protein) y antígenos específicos de Mycobacterium tuberculosis (CFP-10) para el diagnóstico de infección latente de tuberculosis en pacientes con artritis reumatoide.
Materiales y métodos. Se llevó a cabo un estudio transversal analítico en pacientes con artritis reumatoide atendidos en el Hospital Universitario San Vicente Fundación, entre enero y diciembre de 2007, a los cuales se les determinó la producción de IFNγ en respuesta a CFP y CFP-10 en sobrenadantes de cultivos de sangre total, y se correlacionó con la reacción en la prueba de tuberculina.
Además, se estableció el grado de concordancia entre ambas pruebas.
Resultados. Se incluyeron 45 pacientes, de los cuales, 14 (31,1 %) tuvieron un diámetro de induración ≥10 mm (tuberculina positiva), nueve (20 %) produjeron IFNγ en respuesta a CFP-10, y siete fueron positivos para ambas pruebas. La correlación entre las pruebas fue de r=0,53 (IC95%: 0,28-0,72) y la concordancia global entre pruebas fue de 80 %, con un coeficiente kappa de 0,48 (IC95%: 0,20-0,76).
Conclusiones. Solo se observaron dos pacientes con tuberculina positiva y CFP-10 positivo “anérgicos” y se encontraron seis pacientes con tuberculina positiva y CFP-10 negativa “falsos positivos para tuberculina”, lo cual sugiere que la prueba de la tuberculina no es la más adecuada para indicar profilaxis para tuberculosis.
Palabras clave: artritis reumatoide, tuberculosis latente, prueba de tuberculina, interferón gamma, factor de necrosis tumoral alfa/antagonistas e inhibidores.
doi: http://dx.doi.org/10.7705/biomedica.v33i2.694
Rheumatoid arthritis (RA) is a chronic inflammatory disorder that primarily attacks synovial joints, and it affects approximately 1% of the world’s population (1). Rheumatoid arthritis causes pain, impairs functional capability, and reduces patient quality of life. Current treatments with anti-TNF-α monoclonal antibody or soluble TNF-α receptor abrogate joint inflammation and damage (2,3).
Many studies have suggested that the clinical use of anti-TNF-α agents for the treatment of rheumatoid arthritis in humans increases the risk of tuberculosis reactivation (4-6). Therefore, screening for latent infection by Mycobacterium tuberculosis (LTBI) is recommended for patients with rheumatoid arthritis before initiating anti-TNF-α therapy to avoid tuberculosis reactivation (7-9). Traditionally, the tuberculin skin test (TST) has been used to detect LTBI. However, the TST is not specific, and its interpretation is often difficult in individuals vaccinated with Mycobacterium bovis bacillus Calmette-Guérin (BCG) and in individuals who have been exposed to environmental mycobacteria (9). In addition, patients on immunosuppressive therapy and those who are HIV infected or have cancer have a diminished TST response (10). Furthermore, the cut-off value of the TST changes according to the tuberculosis prevalence, BCG coverage and nontuberculosis mycobacteria exposure (11,12).
IFNγ release assays (IGRAs), which measure the T-cell response to the Mycobacterium tuberculosisspecific antigens ESAT-6 and CFP-10 (13-17), are new specific and sensitive alternatives for LTBI diagnosis. Therefore, the management guidelines for LTBI diagnosis in some countries are replacing the TST with IGRAs (14-16), although inconsistent results have been reported (11-14,16,18,19). Our aim was to compare the TST and IFNγ production induced by Mycobacterium tuberculosis (Mtb) CFP and Mtb-specific CFP-10 antigens in rheumatoid arthritis patients in Colombia, a country with an intermediate tuberculosis prevalence (20) and high BCG vaccination coverage (72.7%) (21).
Materials and methods
Patients
A transverse analytic study was conducted. The study included patients older than 18 years of age with rheumatoid arthritis who were diagnosed according to the criteria of the American College of Rheumatology (1987) (22) and had never received biological therapy. The patients were recruited at the Servicio de Reumatología, Hospital Universitario San Vicente Fundación, Medellín, Colombia, between January and December 2007. Patients diagnosed with diabetes, cancer, HIV infection, hepatic or renal disease, or active tuberculosis were excluded. The study was approved by the Ethics Committee of Instituto de Investigaciones Médicas, Facultad de Medicina, Universidad de Antioquia, and all participants signed a consent form.
Clinical evaluation The clinical and therapeutic characteristics of the patients were collected during the medical examination and were registered on a questionnaire designed for the investigation. The results of routine laboratory tests were obtained from the clinical records.
Tuberculin skin test
Patients underwent skin testing by an intradermal injection of 0.1 ml of tuberculin (RT23, Statens Serum Institut, Copenhagen, Denmark) in the left forearm volar surface. All the indurations were measured after 72 hours using the ballpoint-pentechnique. The values were categorized considering a cut-off value >10 mm for a positive test.
Blood collection and cell culture
Before the TST, peripheral blood samples were obtained by venipuncture and collected into EDTA tubes. Whole blood cultures were performed as follows: 20 μl of whole blood was diluted in 180 μl of RPMI-1640 (GIBCO, Grand Island, NY) containing Mtb antigens (CFP 10 μg/ml and CFP-10 5 μg/ ml; kindly provided by J. Belisle and K. Dobos, Colorado State University, Contract number: NIH, NIAID ND1-AI-40091, TB Vaccine Testing and Research Materials Contract) or medium alone. Cultures were incubated for seven days at 37°C in 5% CO2. The supernatants were collected and stored at -70°C (23).
IFN-γ measurement by enzyme-linked immunosorbent assay
The determination of IFNγ released into whole culture supernatant was performed by enzymelinked immunosorbent assay (ELISA) following the protocol recommended by the manufacturer (R&D systems, Minneapolis, MN). Briefly, ELISA plates (MaxiSorp, Nalge Nunc International, Rochester, NY) were incubated overnight at room temperature with 100 μl of diluted IFNγ capture antibody. The plates were then washed 3 times with 300 μl of wash buffer (0.05% Tween-20 in PBS, pH 7.4) and dried by tapping on absorbent paper. Blocking was performed by filling the wells with blocking buffer [PBS containing 1% bovine serum albumin, 5% sucrose and 0.05% NaN3] and incubating the plate for 2 h. After washing, samples and standards were prepared in diluent buffer [0.1% bovine serum albumin, 0.05 % Tween-20 in Tris-buffered saline pH 7.3 (20 mM Trizma base, 150 mM NaCl)], and 100 μl of the supernatant dilutions was added to the wells and incubated μl of diluted biotinylated detection antibody was dispensed into each well, and the plates were incubated for 2 h at room temperature. The anti-IFNγ biotinylated antibody was diluted in 2% heat-inactivated normal goat serum 2 h before use. The plates were then washed and 100 μl of a 1/200 dilution of horseradish peroxidase (HRP)- streptavidin conjugate in Poly-HRP dilution buffer (Endogen, Woburn, MA) was added to each well before incubation for 20 min. The plates were washed, and 100 μl of substrate reagent was dispensed into each well and incubated at room temperature for 12 min in the dark. The reaction was stopped using 50 μl/well of stop solution (0.18 M H2SO4), and optical densities were read immediately at 450 nm and corrected at 490 nm (Bio-Tek Instruments, Kimpton, UK).
Statistical analyses
Absolute and relative frequencies were used to describe qualitative variables, and the median and interquartile range were used for quantitative variables. The normality of the distribution was evaluated using Shapiro-Wilk´s test. The amounts of IFNγ released in response to CFP and CFP- 10 were calculated by interpolation to non-linear regression standard curves. The cut-off value for IFNγ production levels was established at 22 pg/ mL, according to a previous report by our group (22). The values of the induration of the TST were evaluated for a correlation with the IFNγ released in response to CFP and CFP-10 by Spearman`s correlation coefficient, and the concordance between tests was evaluated by the global concordance and Kappa coefficient. Analyses were performed using GraphPad Prism version 5 (GraphPad Software, San Diego, California, USA).
Results
Clinical characteristics of the studied population
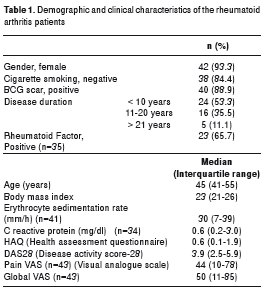
During the study period, 45 patients were selected according to the eligibility criteria. The study included 42 women, and the median age was 45.5 years (interquartile range, 41 to 55 years). A disease duration of less than 10 years was observed in 24 patients (53.3%), and 40 patients (88.9%) had a BCG scar. Most patients had mild to moderate RA activity, which was evaluated with the DAS-28 (24) and acute phase proteins (APs) measured by the sedimentation rate and the C- reactive protein level (table 1).

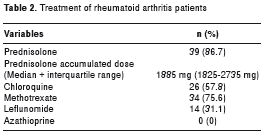
The most frequently used disease-modifying antirheumatic drug (DMARD) was methotrexate in 34 (75.6%) patients, while 39 (86.7%) patients were taking steroids with a previous year median accumulated dose of 1885 mg (1825 mg-2735 mg) (table 2).
Comparison of TST and IFN γ-based blood tests
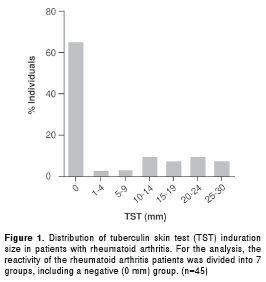
As shown in figure 1, 29 patients (64.4%) had a TST induration of 0 mm, 14 patients (31.1%) had a TST induration ≥ 10 mm, and only 2 (4.4%) patients had a TST induration between 1 and 9 mm.
The production of IFNγ induced by CFP stimuli was ≥ 22 pg/ml in 18 (40%) patients, while only 9 (20%) produced ≥ 22 pg/ml of IFNγ in response to CFP- 10. These 9 patients were considered positive for LTBI, given the high specificity of CFP-10 for TB infection.
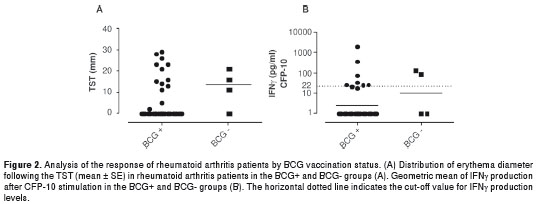
The distribution of the results obtained based on the vaccination status is shown in figure 2. Because the majority of patients had been vaccinated with BCG, it was not possible to determine whether vaccination influenced the TST and IGRA results.
Correlation between the TST and IGRAs in rheumatoid arthritis patients
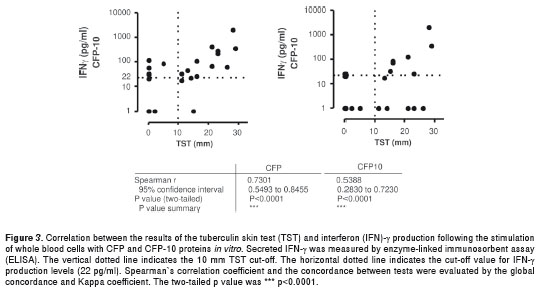
Because the diagnostic value of the TST test has been questioned due to its low specificity in individuals vaccinated with Mycobacterium bovis bacillus Calmette-Guérin (BCG) and individuals exposed to environmental mycobacteria, we evaluated the correlation between this test and the IFNγ response to CFP and CFP-10. Because the data did not follow a normal distribution, Spearman´s correlation test was used. The TST correlated with the CFP- and CFP-10-induced IFNγ levels (Spearman r=0.73 95% CI 0.54-0.84, p<0.0001, and Spearman r=0.53 95%CI 0.28- 0.72, p<0.001, respectively) (figure 3). Six patients (13.3%) presented a TST induration greater than or equal to 10 mm and were CFP10 negative (< 22 pg/ ml IFNγ) and were thus considered false-positive TST patients. In contrast, only two patients (4.4%) had a TST induration < 10 mm and were CFP10 positive (> 22 pg/ml IFNγ) and were considered “anergic”. The global agreement between CFP10 and the TST was 80%, with an expected agreement of 61% and a coefficient κ= 0.48 (CI 95% 0.20- 0.76). The agreement between the TST and CFP was 82%, with an expected agreement of 54% and a coefficient κ= 0.61 (CI 95% 0.38-0.85).
Discussion
TNF-α plays a major role in the immune response against Mtb, and the increase in the TB case number associated with anti-TNF-α therapy has increased its importance (25). The diagnosis and chemoprophylaxis of LTBI is an important issue to consider before initiating anti-TNF therapy in patients with rheumatologic diseases such as rheumatoid arthritis (18,26,27).
The TST has traditionally been used to detect LTBI; however, it is not a very specific diagnostic tool (28,29). This may be explained by the possible effects of BCG vaccination (30-32) and the presence of environmental mycobacteria (33), which can increase the incidence of false-positive results in countries such as Colombia, where vaccination is widely and routinely performed. The number of patients without BCG in our study was very low and did not allow us to determine whether previous vaccination influenced the TST response.
Furthermore, in our study, six patients had a TST induration >10 mm and were CFP10 negative, and due to the higher specificity of the IGRAs, we interpreted these patients as having false-positive TST results. Additionally, as in other previously reported studies of autoimmune diseases such as rheumatoid arthritis and lupus in countries with high vaccination indexes, our study demonstrated a poor concordance between the TST and IGRAs (16,34,35). Given the high specificity of IGRAs, these assays should be used as confirmatory tests of infection by Mtb, particularly in rheumatoid arthritis patients for whom TST screening is positive, to avoid false-positive diagnoses and allow an accurate decision regarding whether to initiate prophylaxis therapy with isoniazid or rifampin.
The effect of immunosuppressive therapy on the TST response should also be considered. The accuracy and reliability of the TST are significantly affected by immunosuppressive therapy and by cellular and humoral immune system disturbances related to RA (13,14,36). In this study, most patients, despite receiving this type of therapy, responded to the TST, and some patients even presented high TST induration values. Therefore, the tuberculin anergy previously described in rheumatoid arthritis patients in other reports (36- 38) was not observed in our study.
The distribution of TST reactivity in our patient cohort showed that 31.1% of patients had a TST induration > 10 mm, whereas only two patients had indurations of 5-9 mm. This finding suggests that a cut-off for TST reactivity of 10 mm may be more appropriate for our patients than the 5 mm cut-off suggested by other studies (39) and the management guidelines of other countries with different TB epidemiologies (7,8).
The significance of TST reactivity is very controversial. Although our sample was small, in contrast to other studies (39), the findings suggest that TST false-positive results are more likely to be observed in rheumatoid arthritis patients than “anergy” and that TST-positive results for LTBI screening in rheumatoid arthritis patients could lead to an overestimation of the proportion of LTBI individuals and should not be used to determine the need for chemoprophylaxis therapy. The main limitation of this study is the small sample size because it was a convenience sample. Therefore, the results must be carefully interpreted in this context. In conclusion, IGRAs are an important second step in the diagnosis of true-positive patients after screening with the TST (40)
Asociación Colombiana de Reumatología, Young Investigator Program of Universidad de Antioquia and Estrategia de Sostenibilidad, Universidad de Antioquia. The antigens were supplied by Colorado State University.
The authors have no conflicts of interest to report regarding the results of this study.
This study was supported by the Asociación Colombiana de Reumatología.
Corresponding author:
Gloria Vásquez, Sede de Investigación Universitaria (SIU), Universidad de Antioquia, Carrera 53 N° 61-30, laboratorio 510, Medellín, Colombia Teléfono: (574) 219 6446; fax: (574) 219 6451 glomavas@gmail.com1. Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010;376:1094-108. http://dx.doi.org/10.1016/S0140-6736(10)60826-4
2. Hochberg MC, Lebwohl MG, Plevy SE, Hobbs KF, Yocum DE. The benefit/risk profile of TNF-blocking agents: Findings of a consensus panel. Semin Arthritis Rheum. 2005;34:819- 36. http://dx.doi.org/10.1016/j.semarthrit.2004.11.006
3. Piguet PF, Grau E, Vesin C, Loetscher H, Gentz R, Lesslauer W. Evolution of collagen arthritis in mice is arrested by treatment with anti-tumour necrosis factor (TNF) antibody or a recombinant soluble TNF receptor. Immunology. 1992;77:510-4.
4. Chan J, Flynn J. The immunological aspects of latency in tuberculosis. Clin Immunol. 2004;110:2-12. http://dx.doi.org/10.1016/S1521-6616(03)00210-9
5. Gardam MA, Keystone EC, Menzies R, Manners S, Skamene E, Long R, et al. Anti-tumour necrosis factor agents and tuberculosis risk: Mechanisms of action and clinical management. Lancet Infect Dis. 2003;3:148-55. http://dx.doi.org/10.1016/S1473-3099(03)00545-0
6. Keane J, Gershon S, Wise RP, Mirabile-Levens E, Kasznica J, Schwieterman WD, et al . Tuberculosis associated with infliximab, a tumor necrosis factor alfa �neutralizing agent. N Engl J Med. 2001;345:1098-104. http://dx.doi.org/10.1056/NEJMoa011110
7. Furst DE, Cush J, Kaufmann S, Siegel J, Kurth R. Preliminary guidelines for diagnosing and treating tuberculosis in patients with rheumatoid arthritis in immunosuppressive trials or being treated with biological agents. Ann Rheum Dis. 2002;61(Suppl.2):ii62-3. http:// dx.doi.org/10.1136/ard.61.suppl_2.ii62
8. Ledingham J, Deighton C; British Society for Rheumatology Standards, Guidelines and Audit Working Group. Update on the British Society for Rheumatology guidelines for prescribing TNF a blockers in adults with rheumatoid arthritis (update of previous guidelines of April 2001). Rheumatology (Oxford). 2005;44:157-63. http://dx.doi.org/10.1093/rheumatology/keh464
9. Tissot F, Zanetti G, Francioli P, Zellweger J-P, Zysset F. Influence of bacille Calmette-Guérin vaccination on size of tuberculin skin test reaction: To what size? Clin Infect Dis. 2005;40211-7. http://dx.doi.org/10.1086/426434
10. Pesanti EL. The negative tuberculin test. Tuberculin, HIV, and anergy panels. Am J Respir Crit Care Med. 1994;149:1699-709.
11. Carmona L, Gómez-Reino JJ, Rodríguez-Valverde V, Montero D, Pascual-Gómez E, Mola EM, et al. Effectiveness of recommendations to prevent reactivation of latent tuberculosis infection in patients treated with tumor necrosis factor antagonists. Arthritis Rheum. 2005;52:1766- 72. http://dx.doi.org/10.1002/art.21043
12. Mariette X, Salmon D. French guidelines for diagnosis and treating latent and active tuberculosis in patients with RA treated with TNF blockers. Ann Rheum Dis. 2003;62:791. http://dx.doi.org/10.1136/ard.62.8.791
13. Arend SM, Engelhard ACF, Groot G, de Boer K, Andersen P, Ottenhoff THM, et al. Tuberculin skin testing compared with t-cell responses to Mycobacterium tuberculosis-specific and nonspecific antigens for detection of latent infection in persons with recent tuberculosis contact. Clin Diagn Lab Immunol. 2001;8:1089-96. http://dx.doi.org/10.1128/CDLI.8.6.1089-1096.2001
14. Beglinger C, Dudler J, Mottet C, Nicod L, Seibold F, Villiger P, et al. Screening for tuberculosis infection before initiation of anti-TNF-a therapy. Swiss Med Wkly. 2007;137:621-2. 15. Nizam S, Emery P. Attenuated response to purified protein derivative in patients with rheumatoid arthritis. Ann Rheum Dis. 2006;65:980. http://dx.doi.org/10.1136/ ard.2005.050161
16. Ponce de León D, Acevedo-Vásquez E, Alvizuri S, Gutiérrez C, Cucho M, Alfaro J, et al. Comparison of an interferon-gamma assay with tuberculin skin testing for detection of tuberculosis (TB) infection in patients with rheumatoid arthritis in a TB-endemic population. J Rheumatol. 2008;35:776-81.
17. Song Q, Guo H, Zhong H, Liu Z, Chen X, Wang C, et al. Evaluation of a new interferon-gamma release assay and comparison to tuberculin skin test during a tuberculosis outbreak. Int J Infect Dis. 2012;16:e522-e6. http://dx.doi. org/10.1016/j.ijid.2012.03.003,
18. Greveson K. Can ELISpot replace the tuberculin skin test for latent tuberculosis? Br J Nurs. 2010;18:1248-54.
19. Schluger NW. Changing approaches to the diagnosis of tuberculosis. American Am J Respir Crit Care Med. 2001;164:2020-4.
20. Organization WH. Global tuberculosis control 2011. Fecha de consulta: 15 de febrero de 2012. Disponible en: http://www.who.int/tb/publications/global_report/en/2011.
21. Ministerio de la Protección Social. Retos y acciones futuras del Programa Ampliado de Inmunizaciones, Colombia 2006-2010. Fecha de consulta: 15 de febrero de 2012. Disponible en: http://www.slideshare.net/malondono4/retospai-min-proteccion-social-2006-2010.
22. Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315-24. http://dx.doi.org/10.1002/art.1780310302
23. Del Corral H, París S, Marín N, Marín D, López L, Henao H, et al. IFNgamma response to Mycobacterium tuberculosis, risk of infection and disease in household contacts of tuberculosis patients in Colombia. PLos One. 2009;4:e8257. http://dx.doi.org/10.1371/journal.pone.0008257
24. Prevoo M, van ´t Hof M, Kuper H, van Leeuwen M, van de Putte L, van Riel P. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995;38:44-8.
25. Wallis RS. Tumour necrosis factor antagonists: Structure, function, and tuberculosis risks. Lancet Infect Dis. 2008;8:601-11. http://dx.doi.org/10.1016/S1473-3099(08)70227-5
26. Lioté H, Lioté F. Role for interferon-gamma release assays in latent tuberculosis screening before TNF- alfa; antagonist therapy. Joint Bone Spine. 2011;78:352-7. http://dx.doi.org/10.1016/j.jbspin.2010.12.001
27. Martin J, Walsh C, Gibbs A, McDonnell T, Fearon U, Keane J, et al . Comparison of interferon ? release assays and conventional screening tests before tumour necrosis factor alfa blockade in patients with inflammatory arthritis. Ann Rheum Dis. 2010;69:181-5.
28. Diel R, Loddesnkemper R, Nienhaus A. Predictive value of interferon- ? release assays and tuberculin skin testing for progression from latent TB infection to disease state: A metaanalysis. Chest. 2012;142:63-75. http://dx.doi.org/10.1378/chest.11-3157.
29. Ramos J, Robledano C, Masiá M, Beldas S, Padilla S, Rodríguez J, et al. Contribution of interferon gamma release assays testing to the diagnosis of latent tuberculosis infection in HIV-infected patients: A comparison of QuantiFERONTB gold in tube, T-SPOT.TB and tuberculin skin test. BMC Infect Dis. 2012;12:169. http://dx.doi.org/10.1186/1471-2334-12-169.
30. Fjällbrant H, Ridell M, Olof larsson L. BCG scar and tuberculin reactivity in children and adults. Scand J Infect Dis.2008;40:387-92. http://dx.doi.org/10.1080/00365540701732905
31. Hernández-Garduño E, Elwood RK. The prevalence and predictors of tuberculin positivity in subjects screened for reasons other than contact investigation. Can Respir J. 2008;15:181-7.
32. Wang L, Turner MO, Elwood RK, Schulzer M, Fitzgerald JM. A meta-analysis of the effect of bacille Calmette Guérin vaccination on tuberculin skin test measurements. Thorax. 2002;57:804-9. 10. http://dx.doi.org/1136/thorax.57.9.804
33. Garzón M, Orjuela D, Naranjo O, Llerena C. Micobacterias no tuberculosas en Colombia 1995-2003. Inf Quinc Epidemiol Nac. 2005;10:161-76.
34. Emery P, Panayi G, Symmons D, Brown G. Mechanisms of depressed delayed-type hypersensitivity in rheumatoid arthritis: The role of protein energy malnutrition. Ann Rheum Dis. 1984;43:430-4.
35. Hsia EC, Schluger N, Cush JJ, Chaisson RE, Matteson EL, Xu S, et al. Interferon-gamma; release assay versus tuberculin skin test prior to treatment with golimumab, a human anti-tumor necrosis factor antibody, in patients with rheumatoid arthritis, psoriatic arthritis, or ankylosing spondylitis. Arthritis Rheum. 2012;64:2068-77. http://dx.doi.org/10.1002/art.34382
36. Yilmaz N, Zehra S, Inanc N, Karakurt S, Direskeneli H, Yavuz S. Comparison of QuantiFERON-TB gold test and tuberculin skin test for the identification of latent Mycobacterium tuberculosis infection in lupus patients.Lupus.2012;21:491-5. http://dx.doi.org/10.1177/0961203311430700
37. Ali M, Ponchel F, Wilson KE, Francis MJD, Wu X, Verhoef A, et al. Rheumatoid arthritis synovial T cells regulate transcription of several genes associated with antigeninduced anergy. J Clin Invest. 2001;107:519-28. http:// dx.doi.org/10.1172/JCI8027
38. Ponchel F, Morgan AW, Bingham SJ, Quinn M, Buch M, Verburg RJ, et al. Dysregulated lymphocyte proliferation and differentiation in patients with rheumatoid arthritis. Blood. 2002;100:4550-6. http://dx.doi.org/10.1182/blood-2002-03-0671
39. Ravelo J, Camargo V, Huamanchumo R, Berrocal A, Calvo A. Respuesta a tuberculina en artritis reumatoide. Un estudio con controles intradomiciliarios. Revista Colombiana de Reumatología. 2005;12:312-9.
40. Chen D-Y, Shen G-H, Hsieh T-Y, Hsieh C-W, Lan J-L. Effectiveness of the combination of a whole-blood interferon-gamma assay and the tuberculin skin test in detecting latent tuberculosis infection in rheumatoid arthritis patients receiving adalimumab therapy. Arthritis Rheum. 2008;59:800-6. http://dx.doi.org/10.1002/art.23705