Las células mesenquimales del estroma olfatorio humano coexpresan proteínas de las células basales horizontales y de recubrimiento neural en cultivo.
Resumen
Introducción. El recambio celular del neuroepitelio olfatorio ocurre durante la vida del individuo gracias a precursores olfatorios. Además, las células mesenquimales del estroma también contribuyen a la homeostasis de la mucosa. Cuando un explante de una biopsia de mucosa se cultiva en un medio esencial mínimo, se genera una población predominante de células adherentes que expresan proteínas típicas de las células mesenquimales del estroma. La coexpresión de marcadores fenotípicos de precursores olfatorios y de células del recubrimiento del nervio olfatorio constituiría una propiedad única de las células mesenquimales del estroma.
Objetivo. Determinar si la diferenciación celular de las células mesenquimales hacia fenotipos neurales modula la expresión de los marcadores mesenquimales característicos.
Materiales y métodos. Se compararon las células aisladas de la mucosa olfatoria en un medio de cultivo con suplemento de 10 % de suero fetal bovino con esferas generadas en un medio sin suero más factores de crecimiento.
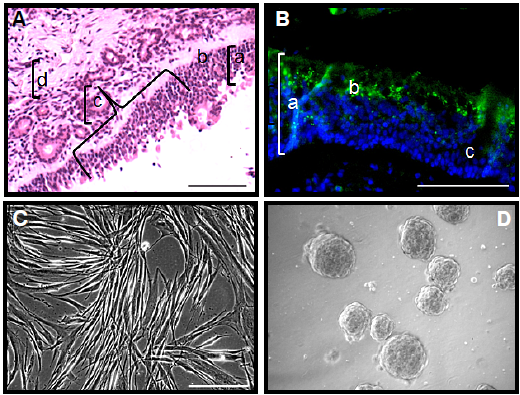
Resultados. Se determinó la expresión de proteínas de las células mesenquimales del estroma (CD29+, CD73+, CD90+, CD45-), de las basales horizontales (ICAM-1/CD54+, p63+, p75NGFr+), y de las del recubrimiento del nervio olfatorio (nestin+, GFAP+) en la misma población cultivada. La determinación de Oct 3/4, Sox-2 y Mash-1, así como de las neurotrofinas BDNF, NT3 y NT4, sugirió que las células del estroma son funcionales. La expresión de las proteínas de las células mesenquimales y los precursores olfatorios, disminuyó en las células de las mesenesferas inducidas por ausencia de suero en el medio de cultivo.
Conclusión. Las células mesenquimales del estroma de la mucosa olfatoria presentan una tendencia dominante hacia la diferenciación neural.
Descargas
Referencias bibliográficas
Schwob JE, Jang W, Holbrook EH, Holbrook EH, Lin B, Herrick DB, et al. Stem and progenitor cells of the mammalian olfactory epithelium: Taking poietic license. J Comp Neurol. 2017;525:1034-54. https://doi.org/10.1002/cne.24105
Schwob JE, Jang W, Holbrook EH. Stem cells of the olfactory epithelium. In: Rao MS, editor. Neural development and stem cells. 3rd edition. New York: Springer Science Business Media; 2012. p. 201-22.
Graziadei PP. Cell dynamics in the olfactory mucosa. Tissue Cell. 1973;5:113-31.
Jang W, Youngentob SL, Schwob JE. Globose basal cells required for reconstitution of olfactory epithelium after methyl bromide lesion. J Comp Neurol 2003;460:123-40. https://doi.org/10.1002/cne.10642
Carter LA, MacDonald JL, Roskams AJ. Olfactory horizontal basal cells demonstrate a conserved multipotent progenitor phenotype. J Neurosci. 2004;24:5670-83. https://doi.org/10.1523/JNEUROSCI.0330-04.2004
Fletcher RB, Prasol MS, Estrada J, Baudhuin A, Vranizan K, Choi YG, et al. p63 regulates olfactory stem cell self-renewal and differentiation. Neuron. 2011;72:748-59. https://doi.org/10.1016/j.neuron.2011.09.009
Holbrook EH, Wu E, Curry WT, Lin DT, Schwob JE. Immunohistochemical characterization of human olfactory tissue. Laryngoscope. 2011;121:1687-701. https://doi.org/10.1002/lary.21856
Deng J, Petersen BE, Steindler DA, Jorgensen ML, Laywell ED. Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells. 2006;24:1054-64. https://doi.org/10.1634/stemcells.2005-0370
Chase LG, Lakshmipathy U, Solchaga LA, Rao MS, Vemuri MC. A novel medium for the expansion of human mesenchymal stem cells. Stem Cell Res Ther. 2010;1:8. https://doi.org/10.1186/scrt8
Delorme B, Nivet E, Gaillard J, Häupl T, Ringe J, Devèze A, et al. The human nose harbors a niche of olfactory ectomesenchymal stem cells displaying neurogenic and osteogenic properties. Stem Cells Dev. 2010;19:853-66. https://doi.org/10.1089/scd.2009.0267
Lindsay SL, Riddell JS, Barnett SC. Olfactory mucosa for transplant-mediated repair: A complex tissue for a complex injury? Glia. 2010;58:125-34. https://doi.org/10.1002/glia.20917
Lindsay SL, Johnstone SA, Mountford JC, Sheikh S, Allan DB, Clark L, et al. Human mesenchymal stem cells isolated from olfactory biopsies but not bone enhance CNS myelination in vitro. Glia. 2013;61:368-82. https://doi.org/10.1002/glia.22440
Fernández VB, Romaniuk MA, Choi H, Labovsky V, Otaegui J, Chasseing NA. Mesenchymal stem cells and their use in therapy: What has been achieved? Differentiation. 2013;85:1-10. https://doi.org/10.1016/j.diff.2012.08.004
Lindsay SL, Toft A, Griffin J, M M Emraja A, Barnett SC, Riddell JS. Human olfactory mesenchymal stromal cell transplants promote remyelination and earlier improvement in gait co-ordination after spinal cord injury. Glia. 2017;65:639-56. https://doi.org/10.1002/glia.23117
Girard SD, Devéze A, Nivet E, Gepner B, Roman FS, Féron F. Isolating nasal olfactory stem cells from rodents or humans. J Vis Exp. 2011;54:1-5. https://doi.org/10.3791/2762
Hahn CG, Han LY, Rawson NE, Mirza N, Borgmann-Winter K, Lenox RH, et al. In vivo and in vitro neurogenesis in human olfactory epithelium. J Comp Neurol. 2005;483:154-63. https://doi.org/10.1002/cne.20424
Heng BC, Cao T, Stanton LW, Robson P, Olsen B. Strategies for directing the differentiation of stem cells into the osteogenic lineage in vitro. J Bone Miner Res 2004;19:1379-94. https://doi.org/10.1359/JBMR.040714
Hess DC, Borlongan CV. Stem cells and neurological diseases. Cell Prolif. 2007;41(Suppl.1):94-114. https://doi.org/10.1111/j.1365-2184.2008.00486.x
Marshall CT, Lu C, Winstead W, Zhang X, Winstead W, Zhang X, et al. The therapeutic potential of human olfactory-derived stem cells. Histol Histopathol. 2006;21:633-43. https://doi.org/10.14670/HH-21.633
Leung CT, Coulombe PA, Reed RR. Contribution of olfactory neural stem cells to tissue maintenance and regeneration. Nat Neurosci. 2007;10:720-6. https://doi.org/10.1038/nn1882
Nivet E, Vignes M, Girard SD, Pierrisnard C, Baril N, Devèze A, et al. Engraftment of human nasal olfactory stem cells restores neuroplasticity in mice with hippocampal lesions. J Clin Invest. 2011;121:2808-20. https://doi.org/10.1172/JCI44489
Ramón-Cueto A, Cordero MI, Santos-Benito FF, Ávila J. Functional recovery of paraplegic rats and motor axon regeneration in their spinal cords by olfactory ensheathing glia. Neuron. 2000;25:425-35. https://doi.org/10.1016/S0896-6273(00)80905-8
Ayala-Grosso CA, Pieruzzini R, Diaz-Solano D, Wittig O, Abrante L, Vargas L, et al. Amyloid-aβ peptide in olfactory mucosa and mesenchymal stromal cells of mild cognitive impairment and Alzheimer’s disease patients. Brain Pathol. 2015;25:136-45. https://doi.org/10.1111/bpa.12169
Solís-Chagoyán H, Flores-Soto E, Reyes-García J, Valdés-Tovar M, Calixto E, Montaño LM, et al. Voltage-activated calcium channels as functional markers of mature neurons in human olfactory neuroepithelial cells: Implications for the study of neurodevelopment in neuropsychiatric disorders. Int J Mol Sci. 2016;17. https://doi.org/10.3390/ijms17060941
Cercós MG, Galván-Arrieta T, Valdés-Tovar M, Solís-Chagoyán H, Argueta J, Benítez-King G, et al. Abnormally increased secretion in olfactory neuronal precursors from a case of schizophrenia is modulated by melatonin: A pilot study. Int J Mol Sci. 2017;18. https://doi.org/10.3390/ijms18071439
Galván-Arrieta T, Trueta C, Cercós MG, Valdés-Tovar M, Alarcón S, Oikawa J, et al. The role of melatonin in the neurodevelopmental etiology of schizophrenia: A study in human olfactory neuronal precursors. J Pineal Res. 2017;63. https://doi.org/10.1111/jpi.12421
Lavoie J, Gassó Astorga P, Segal-Gavish H, Wu YC, Chung Y, Cascella NG, et al. The olfactory neural epithelium as a tool in neuroscience. Trends Mol Med. 2017;23:100-3. https://doi.org/10.1016/j.molmed.2016.12.010
Masurkar AV, Devanand DP. Olfactory dysfunction in the elderly: Basic circuitry and alterations with normal aging and Alzheimer’s disease. Curr Geriatr Rep. 2014;3:91-100. https://doi.org/10.1007/s13670-014-0080-y
Jiménez-Vaca AL, Benítez-King G, Ruiz V, Ramírez-Rodríguez GB, Hernández-de la Cruz B, Salamanca-Gómez FA, et al. Exfoliated human olfactory neuroepithelium: A source of neural progenitor cells. Mol Neurobiol. 2018;55:2516-23. https://doi.org/10.1007/s12035-017-0500-z
Doty RL. Olfactory dysfunction in Parkinson disease. Nat Rev Neurol. 2012;8:329-39. https://doi.org/10.1038/nrneurol.2012.80
Murrell W, Féron F, Wetzig A, Cameron N, Splatt K, Bellette B, et al. Multipotent stem cells from adult olfactory mucosa. Dev Dyn. 2005;233:496-515. https://doi.org/10.1002/dvdy.20360
Zhang X, Klueber KM, Guo Z, Lu C, Roisen FJ. Adult human olfactory neural progenitors cultured in defined medium. Exp Neurol. 2004;186:112-23. https://doi.org/10.1016/j.expneurol.2003.10.022
Lovell MA, Jafek BW, Moran DT, Rowley JC 3rd. Biopsy of human olfactory mucosa. An instrument and a technique. Arch Otolaryngol. 1982;108:247-9.
Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem.1997;64:295-312.
Murdoch B, Roskams AJ. Olfactory epithelium progenitors: Insights from transgenic mice and in vitro biology. J Mol Histol. 2007;38:581-99. https://doi.org/10.1007/s10735-007-9141-2
Wislet-Gendebien S, Bruyère F, Hans G, Leprince P, Moonen G, Rogister B. Nestin-positive mesenchymal stem cells favour the astroglial lineage in neural progenitors and stem cells by releasing active BMP4. BMC Neurosci. 2004;5:33. https://doi.org/10.1186/1471-2202-5-33
Wislet-Gendebien S, Leprince P, Moonen G, Rogister B. Regulation of neural markers nestin and GFAP expression by cultivated bone marrow stromal cells. J Cell Sci. 2003;116:3295-302. https://doi.org/10.1242/jcs.00639
Mark P, Kleinsorge M, Gaebel R, Lux CA, Toelk A, Pittermann E, et al. Human mesenchymal stem cells display reduced expression of CD105 after culture in medium. Stem Cells Int. 2013;2013:698076. https://doi.org/10.1155/2013/698076
Othman M, Lu C, Klueber K, Winstead W, Roisen F. Clonal analysis of adult human olfactory neurosphere forming cells. Biotech Histochem. 2005;80:189-200. https://doi.org/10.1080/10520290500469777
Krolewski RC, Jang W, Schwob JE. The generation of olfactory epithelial neurospheres in vitro predicts engraftment capacity following transplantation in vivo. Exp Neurol. 2011;229:308-23. https://doi.org/10.1016/j.expneurol.2011.02.014
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























