Metacyclogenesis of Trypanosoma cruzi in B. ferroae (Reduviidae: Triatominae) and feces infectivity under laboratory conditions
Abstract
Introduction: Belminus ferroae is a triatominae with entomophagous behavior. However, it may occasionally feed on vertebrates. Currently, there is no evidence of natural infection with Trypanosoma cruzi or the occurrence of metacyclogenesis in this species.
Objective: To test T. cruzi metacyclogenesis in B. ferroae and the infectivity of their feces or intestinal contents in rodents under laboratory conditions.
Materials and methods: Twenty nymphs of B. ferroae were infected with an autochthonous strain of T. cruzi (M/HOM/VE/09/P6). Fecal and urine samples were collected from spontaneous droppings or by compressing the bugs’ abdomens and, eventually, by removing their gut contents, and then examined at 10, 20, 30, 40, 50, and 60 days. We quantified T. cruzi parasitic load, as well as the evolutionary forms in feces, urine, and intestinal contents by Giemsa staining. Similarly, we evaluated the infectivity of T. cruzi metacyclic trypomastigotes in albino mice.
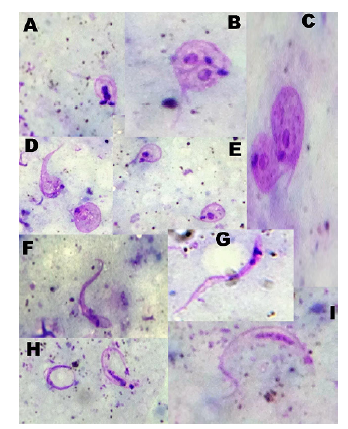
Results: The parasitological analysis showed three insects (15%) infected with T. cruzi at 30 (n=1), 40 (n=1), and 50 (n=1) days post-infection. We observed parasitic loads of up to 1.62 x 105 trypanosomes/mm3 and metacyclogenesis percentages between 3.5% and 6.78%.
Conclusions: This is the first time that T. cruzi metacyclogenesis is reported in a species of the genus Belminus under laboratory conditions and the infectivity of Belminus’ feces is demonstrated on a vertebrate host.
Downloads
References
Otálora-Luna F, Pérez-Sánchez AJ, Sandoval C, Aldana E. Evolution of hematophagous habit in Triatominae (Heteroptera: Reduviidae). Rev Chil Hist Nat. 2015;88:1-13. https://doi.org/10.1186/S40693-014-0032-0
Stanaway JD, Roth G. The burden of chagas disease estimates and challenges. Glob Heart. 2015;10:139-44. https://doi.org/10.1016/j.gheart.2015.06.001
Lent H, Wygodzinsky P. Revision of the Triatominae (Hemiptera, Reduviidae), and their significance as vectors of Chagas disease. Bull Am museum Nat Hist. 1979;163:123-520.
Schofield CJ, Galvão C. Classification, evolution, and species groups within the Triatominae. Acta Trop. 2009;110:88-100. https://doi.org/10.1016/j.actatropica.2009.01.010
Sandoval CM, Joya MI, Gutiérrez R, Angulo VM. Cleptohaematophagy of the Triatomine bug Belminus herreri. Med Vet Entomol. 2000;14:100-1. https://doi.org/10.1046/j.1365-2915.2000.00210
Sandoval CM, Medone P, Nieves EE, Jaimes DA, Ortiz N, Rabinovich JE. Demographic fitness of Belminus ferroae (Hemiptera: Triatominae) on three different hosts under laboratory conditions. Mem Inst Oswaldo Cruz. 2013;108:854-64. https://doi.org/10.1590/0074-0276130211
Díaz-Albiter HM, Ferreira TN, Costa SG, Rivas GB, Gumiel M, Cavalcante DR, et al. Everybody loves sugar: First report of plant feeding in triatomines. Parasit Vectors. 2016;9. https://doi.org/10.1186/s13071-016-1401-0
Durán P, Siñani E, Depickère S. On triatomines, cockroaches and haemolymphagy under laboratory conditions: New discoveries. Mem Inst Oswaldo Cruz. 2016;111:605-13. https://doi.org/10.1590/0074-02760160027
Schmidt JO, Dorn PL, Klotz SA. Second-best is better than nothing: Cockroaches as a viable food source for the kissing bug Triatoma recurva (Hemiptera: Reduviidae). J Med Entomol. 2019;56:651-5. https://doi.org/10.1093/jme/tjy233
Sandoval CM, Duarte R, Gutiérrez R, Da Silva Rocha D, Angulo VM, Esteban L, et al. Feeding sources and natural infection of Belminus herreri (Hemiptera, Reduviidae, Triatominae) from dwellings in Cesar, Colombia. Mem Inst Oswaldo Cruz. 2004;99:137-40. https://doi.org/10.1590/S0074-02762004000200004
Sandoval CM, Ortiz N, Jaimes D, Lorosa E, Galvão C, Rodríguez O, et al. Feeding behaviour of Belminus ferroae (Hemiptera: Reduviidae), a predaceous Triatominae colonizing rural houses in Norte de Santander, Colombia. Med Vet Entomol. 2010;24:124-31. https://doi.org/10.1111/j.1365-2915.2010.00868.x
Sandoval CM, Nieves EE, Gutiérrez R, Jaimes DA, Rodríguez NO, Otálora-Luna F, et al. Morphometric analysis of the host effect on phenotypical variation of Belminus ferroae (Hemiptera: Triatominae). Psyche. 2015;2015. https://doi.org/10.1155/2015/613614
Gil-Santana H. Estudo taxonômico da tribo Bolboderini (Hemiptera-Heteroptera, Reduviidae, Triatominae), com análise cladística. Rio de Janeiro: Instituto Oswaldo Cruz; 2014. Fecha de consulta: 21 de mayo de 2020. Disponible en: https://www.arca.fiocruz.br/handle/icict/12124
Galvão C, Angulo VM. Belminus corredori, a new species of Bolboderini (Hemiptera: Reduviidae: Triatominae) from Santander, Colombia. Zootaxa. 2006;1241:61-8. https://doi.org/10.11646/zootaxa.1241.1.4
Sandoval CM. Aspectos biológicos y morfológicos de Belminus sp. de Norte de Santander, Colombia. Mérida: Universidad de Los Andes; 2007.
Alarcón M, Pérez MC, Villarreal J, Araújo S, Goncalves L, González A, et al. Detección de ADN de Trypanosoma cruzi en la placenta y fetos de ratones con infección chagásica aguda. Invest Clin. 2009;50:335-45.
Yarbuh AL, Cáceres K, Sulbarán D, Araujo S, Moreno E, Carrasco HJ, et al. Proliferación de Trypanosoma cruzi en la membrana peritoneal y líquido ascítico de ratones con infección aguda. Bol Malariol y Salud Ambient. 2013;53:146-56.
Brenner Z. Observações sobre a inmunidade a superinfecções em camundongos experimentalmente inoculados com Trypanosoma cruzi e submetidos a tratamento. Rev Inst Med Trop São Paulo.1962;4:119-23.
Perlowagora-Szumlewicz A, Moreira CJ. In vivo differentiation of Trypanosoma cruzi-1. Experimental evidence of the influence of vector species on metacyclogenesis. Mem Inst Oswaldo Cruz. 1994;89:603-18. https://doi.org/10.1590/s0074-02761994000400018
Suárez-Quevedo Y, Barbosa-Vinasco HM, Gutiérrez-Garnizo SA, Olaya-Morales JL, Zabala-González D, Carranza-Martínez JL, et al. Innate trypanolytic factors in triatomine hemolymph against Trypanosoma rangeli and T. cruzi: A comparative study in eight Chagas disease vectors. Rev Acad Colomb Cienc Ex Fis Nat. 2020;44:88-104. https://doi.org/10.18257/raccefyn.1097
Basseri HR, Dadi-Khoeni A, Bakhtiari R, Abolhassani M, Hajihosseini-Baghdadabadi R. Isolation and purification of an antibacterial protein from immune induced haemolymph of American Cockroach, Periplaneta americana. J Arthropod Borne Dis. 2016;10:519-27.
Some similar items:
- Víctor Manuel Angulo, Lyda Esteban, Katherine Paola Luna, Attalea butyracea palms adjacent to housing as a source of infestation by Rhodnius prolixus (Hemiptera: Reduviidae) , Biomedica: Vol. 32 No. 2 (2012)
- Cielo León, Mario Iván Ortiz, Catalina Tovar, Jorge Negrete, Ernesto Arroyo, Camila González, Detection of Trypanosoma cruzi strains circulating in Córdoba department (Colombia) isolated from triatomines (Hemiptera: Reduviidae) collected by the community , Biomedica: Vol. 39 No. 2 (2019)
- Lizbeth Díaz, Karen Covarrubias, Ángel Licón, Mixtli Astorga, Yaneth Moreno, José Alejandro Martínez, Biological parameters of Meccus phyllosomus phyllosomus (Burmeister), 1835, Triatoma recurva (Stål), 1868 (Hemiptera, Reduviidae) and their laboratory hybrids , Biomedica: Vol. 37 No. Sup. 2 (2017): Suplemento 2, Entomología médica, 2017
- María Clara Echeverry, Nubia Catalina Tovar, Guillermo Mora, Presence of antibodies to cardiac neuroreceptors in patients with Chagas disease , Biomedica: Vol. 29 No. 3 (2009)
- Marlene Reyes, Víctor Manuel Angulo, Life cycle of Triatoma dimidiata Latreille, 1811 (Hemiptera, Reduviidae) under laboratory conditions: production of nymphs for biological tests , Biomedica: Vol. 29 No. 1 (2009)
- Concepción Judith Puerta, Johana María Guevara, Paula Ximena Pavía, Marleny Montilla, Rubén Santiago Nicholls, Edgar Parra, Yuli Katherine Barrera, Evaluation of TcH2AF-R and S35-S36 primers in PCR tests for the detection of Trypanosoma cruzi in mouse cardiac tissue , Biomedica: Vol. 28 No. 4 (2008)
- Dairo Alonso Rendón, Carlos M. Genes, Omar Triana, Myocardial cellular damage and the activity of the mitochondrial ATP synthase in rats infected with a Colombian strain of Trypanosoma cruzi , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Sandra Paola Santander, Adriana Cuéllar, María del Carmen Thomas, Fanny Guzmán, Alberto Gómez, Manuel Carlos López, Concepción Puerta, Expression of markers on dendritic cells from chronic chagasic patients stimulated with the KMP-11 protein and the K1 peptide from Trypanosoma cruzi , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- José Alejandro Martínez-Ibarra, Jorge Alejandro Martínez-Grant, Miguel Roberto Verdugo-Cervantes, Rafael Bustos-Saldaña, Benjamín Nogueda-Torres, Monitoring triatomid bug (Hemiptera: Reduviidae) presence by sentinel chicken coops in Southern Jalisco State, México , Biomedica: Vol. 30 No. 1 (2010)
- Nelson Grisales, Omar Triana, Víctor Angulo, Nicolás Jaramillo, Gabriel Parra-Henao, Francisco Panzera, Andrés Gómez-Palacio, Genetic differentiation of three Colombian populations of Triatoma dimidiata (Heteroptera: Reduviidae) by ND4 mitochondrial gene molecular analysis , Biomedica: Vol. 30 No. 2 (2010)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























