Disagreement between PCR and serological diagnosis of Trypanosoma cruzi infection in blood donors from a Colombian endemic region
Abstract
Introduction: Chagas’ disease is the leading cause of infectious myocarditis worldwide. This infection caused by Trypanosoma cruzi is usually life-long and asymptomatic; however, the third part of infected people can develop severe or even fatal cardiomyopathy. As the parasitemia in the chronic phase is both low-grade and intermittent, T. cruzi infection is principally detected by serology, although this method has sensitivity and specificity limitations.
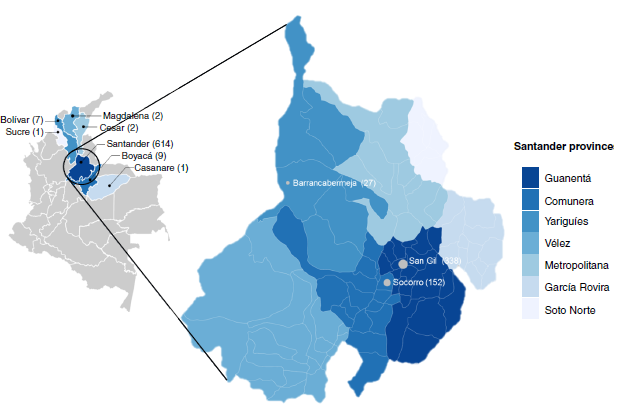
Objective: To determine the level of agreement between serologic and molecular tests in 658 voluntary blood donors from six provinces in the Colombian department of Santander.
Materials and methods: We evaluated an array of diagnostic technologies by cross-section sampling performing a serological double diagnostic test for T. cruzi antibody detection (Chagas III ELISA™, BiosChile Group, and ARCHITECT Chagas CMIA™, Abbott), and DNA detection by polymerase chain reaction (PCR). We collected the demographic, clinical, and epidemiological information of participants. The sample size was calculated using Epidat™ and the statistical analysis was done with Stata 12.1™.
Results: PCR was six times more sensitive in detecting T. cruzi infection than ELISA/CMIA with prevalence values of 1.8% (12/658) and 0.3% (2/658), respectively, and kappa=0.28 (95%CI: -0.03 - 0.59). In contrast, serology showed a sensitivity of 16.7% (95%CI: 2.09 - 48.4) and a specificity of 100% (95%CI: 99.4 - 100). All seropositive samples were found to be positive by PCR.
Conclusions: The implementation of PCR as a complementary method for screening donors could reduce the probability of false negative and the consequent risk of transfusional-transmission of Chagas’ disease, especially in endemic regions.
Downloads
References
World Health Organization. Investing to overcome the global impact of neglected tropical diseases. Third WHO report on neglected tropical diseases. February, 2015. Accessed: March 19, 2018. Available at: https://apps.who.int/iris/bitstream/handle/10665/152781/9789241564861_eng.pdf;jsessionid=404097E9D1CA2695A8D268A0464A0B12?sequence=1
Hotez PJ, Dumonteil E, Heffernan MJ, Bottazzi ME. Innovation for the ‘bottom 100 million’: Eliminating neglected tropical diseases in the Americas. Adv Exp Med Biol. 2013;764:1-12. https://doi.org/10.1007/978-1-4614-4726-9_1
Gunda R, Chimbari M, Mukaratirwa S. Assessment of burden of malaria in Gwanda District, Zimbabwe, using the disability adjusted life years. Int J Environ Res Public Health. 2016;13:244. https://doi.org/10.3390/ijerph13020244
Robertson LJ, Devleesschauwer B, De Noya BA, González ON, Torgerson PR. Trypanosoma cruzi: Time for international recognition as a foodborne parasite. PLoS Negl Trop Dis. 2016;10:e0004656. https://doi.org/10.1371/journal.pntd.0004656
Instituto Nacional de Salud. Sivigila, República de Colombia. Accessed: May 16, 2017. Available at: https://www.ins.gov.co/Direcciones/Vigilancia/Paginas/SIVIGILA.aspx
Morillo CA, Marin-Neto JA, Avezum A, Sosa-Estani S, Rassi Jr A, Rosas F, et al. Randomized trial of benznidazole for chronic Chagas’ cardiomyopathy. N Eng J Med. 2015;373:1295-306. https://doi.org/10.1056/NEJMoa1507574
Ramsey JM, Elizondo-Cano M, Sánchez-González G, Peña-Nieves A, Figueroa-Lara A. Opportunity cost for early treatment of Chagas disease in México. PLoS Negl Trop Dis. 2014;8:e2776. https://doi.org/10.1371/journal.pntd.0002776
Buss LF, De Oliveira-da Silva LC, Moreira CH, Manuli ER, Sales FC, Morales I, et al. Declining antibody levels to Trypanosoma cruzi correlate with polymerase chain reaction positivity and electrocardiographic changes in a retrospective cohort of untreated Brazilian blood donors. PLoS Negl Trop Dis. 2020;14:e0008787. https://doi.org/10.1371/journal.pntd.0008787
Pan American Health Organization. Guidelines for the diagnosis and treatment of Chagas disease. 2019. Accessed: December 7, 2020. Available at: https://iris.paho.org/bitstream/handle/10665.2/49653/9789275120439_eng.pdf?sequence=6&isAllowed=y
Abras A, Gállego M, Llovet T, Tebar S, Herrero M, Berenguer P, et al. Serological diagnosis of chronic Chagas disease: Is it time for a change? J Clin Microbiol. 2016;54:1566-72. https://doi.org/10.1128/JCM.00142-16
Marcon GE, Andrade PD, De Albuquerque DM, Wanderley JS, De Almeida EA, Guariento ME, et al. Use of a nested polymerase chain reaction (N-PCR) to detect Trypanosoma cruzi in blood samples from chronic Chagasic patients and patients with doubtful serologies. Diagn Microbiol Infect Dis. 2002;43:39-43. https://doi.org/10.1016/S0732-8893(02)00366-8
Salomone OA, Basquiera AL, Sembaj A, Aguerri AM, Reyes ME, Omelianuk M, et al. Trypanosoma cruzi in persons without serologic evidence of disease, Argentina. Emerg Infect Dis. 2003;9:1558-62. https://doi.org/10.3201/eid0912.030008
Batista AM, Aguiar C, Almeida EA, Guariento ME, Wanderley JS, Costa SC. Evidence of Chagas disease in seronegative Brazilian patients with megaesophagus. Int J Infect Dis. 2010;14:974-7. https://doi.org/10.1016/j.ijid.2010.05.017
Alarcón B, Colmenares C, Díaz-Bello Z, Ruiz-Guevara R, Medina K, Muñoz-Calderón A, et al. Orally-transmitted Chagas disease: Epidemiological, clinical, serological and molecular outcomes of a school microepidemic in Chichiriviche de la Costa, Venezuela. Parasite Epidemiol Control. 2016;1:188-98. https://doi.org/10.1016/j.parepi.2016.02.005
Sabino EC, Lee TH, Montalvo L, Nguyen ML, Leiby DA, Carrick DM, et al. Antibody levels correlate with detection of Trypanosoma cruzi DNA by sensitive polymerase chain reaction assays in seropositive blood donors and possible resolution of infection over time. Transfusion. 2013;53:1257-65. https://doi.org/10.1111/j.1537-2995.2012.03902.x
Moreira OC, Ramírez JD, Velázquez E, Dias-Melo MF, Lima-Ferreira C, Guhl F, et al. Towards the establishment of a consensus real-time qPCR to monitor Trypanosoma cruzi parasitemia in patients with chronic Chagas disease cardiomyopathy: A substudy from the BENEFIT trial. Acta Trop. 2013;125:23-31. https://doi.org/10.1016/j.actatropica.2012.08.020
Schijman AG, Bisio M, Orellana L, Sued M, Duffy T, Mejía-Jaramillo AM, et al. International study to evaluate PCR methods for detection of Trypanosoma cruzi DNA in blood samples from Chagas disease patients. PLoS Negl Trop Dis. 2011;5:e931. https://doi.org/10.1371/journal.pntd.0000931
Remesar M, Sabino EC, Del Pozo A, Mayer A, Busch MP, Custer B. Bimodal distribution of Trypanosoma cruzi antibody levels in blood donors from a highly endemic area of Argentina: What is the significance of low-reactive samples? Transfusion. 2015;55:2499-504. https://doi.org/10.1111/trf.13180
Chevillard C, Silva JP, Frade AF, Almeida RR, Pandey RP, Nascimento MS, et al. Disease tolerance and pathogen resistance genes may underlie Trypanosoma cruzi persistence and differential progression to Chagas disease cardiomyopathy. Front Immunol. 2018;9:2791. https://doi.org/10.3389/fimmu.2018.02791
Teixeira AR, Nascimento RJ, Sturm NR. Evolution and pathology in Chagas disease: A review. Mem Inst Oswaldo Cruz. 2006;101:463-91. https://doi.org/10.1590/S0074-02762006000500001
Sturm NR, Degrave W, Morel C, Simpson L. Sensitive detection and schizodeme classification of Trypanosoma cruzi cells by amplification of kinetoplast minicircle DNA sequences: Use in diagnosis of Chagas’ disease. Mol Biochem Parasitol. 1989;33:205-14. https://doi.org/10.1016/0166-6851(89)90082-0
Schmunis GA. Trypanosoma cruzi, the etiologic agent of Chagas’ disease: Status in the blood supply in endemic and nonendemic countries. Transfusion. 1991;31:547-57. https://doi.org/10.1046/j.1537-2995.1991.31691306255.x
Behrend M, Beltrán M, Restrepo M, Kroeger A. Control de la enfermedad de Chagas en bancos de sangre de Colombia. Biomédica. 2002;22:39-45. https://doi.org/10.7705/biomedica.v22i1.1138
Rocha LP, Hernández IP, Martínez DM, Castellanos YZ. Trypanosoma cruzi: prevalencia y factores de riesgo de seropositividad en donantes de sangre del Hemocentro y Unidad de Aféresis, Valledupar, Colombia, 2013-2014. Med Lab. 2014;20:529-42.
Flórez C, Guasmayan L, Cortés L, Caicedo A, Beltrán M, Muñoz L. Chagas disease and its seroprevalence in three departments of the Colombian Amazon. Nova. 2016;14:35-43.
Beltrán M, Hilarión LB, Berrío M, Bermúdez MI. Detección de anticuerpos para Trypanosoma cruzi en donantes de sangre. Caquetá, Colombia, 1995 a 2010. Rev Salud Pública. 2017;19:355-61. https://doi.org/10.15446/rsap.v19n3.49627
Moncayo A, Silveira AC. Current epidemiological trends for Chagas disease in Latin America and future challenges in epidemiology, surveillance and health policy. Mem Inst Oswaldo Cruz. 2009;104:17-30. https://doi.org/10.1590/s0074-02762009000900005
Daza N, Sánchez M, Vanegas T, Ortega I. Prevalencia de infecciones en donantes de sangre en la Universidad Industrial de Santander versus parques de la ciudad de Bucaramanga, 2014. Médicas UIS. 2016;29:55-60. https://doi.org/10.18273/revmed.v29n3-2016006
Flores M, Cruz I, Rodríguez M, Nieto J, Franco E, Gárate T, et al. Comparación de técnicas serológicas convencionales y no convencionales para el diagnóstico de la enfermedad de Chagas importada en España. Enferm Infecc Microbiol Clin. 2010;28:284-93. https://doi.org/10.1016/j.eimc.2009.07.005
Castellanos YZ, Cucunubá ZM, Flórez AC, Orozco-Vargas LC. Reproducibilidad de pruebas serológicas para el diagnóstico de infección por Trypanosoma cruzi en mujeres embarazadas de una zona endémica de Santander, Colombia. Biomédica. 2014;34:198-206. https://doi.org/10.7705/biomedica.v34i2.1571
Gomes ML, Galvao LM, Macedo AM, Pena SD, Chiari E. Chagas’ disease diagnosis: Comparative analysis of parasitologic, molecular, and serologic methods. Am J Trop Med Hyg. 1999;60:205-10. https://doi.org/10.4269/ajtmh.1999.60.205
Gutiérrez R, Angulo VM, Tarazona Z, Britto C, Fernandes O. Comparison of four serological tests for the diagnosis of Chagas disease in a Colombian endemic area. Parasitology. 2004;129:439-44. https://doi.org/10.1017/S0031182004005785
Reyes M, Torres Á, Esteban L, Flórez M, Angulo VM. Risk of transmission of Chagas disease by intrusion of triatomines and wild mammals in Bucaramanga, Santander, Colombia. Biomédica. 2017;37:68-78. https://doi.org/10.7705/biomedica.v37i1.3051
Eiro C, Melisa D, Álvarez MG, Cooley G, Viotti RJ, Bertocchi GL, et al. The significance of discordant serology in Chagas disease: Enhanced T-cell immunity to Trypanosoma cruzi in serodiscordant subjects. Front Immunol. 2017;8:1141. https://doi.org/10.3389/fimmu.2017.01141
Bréniere SF, Poch O, Selaes H, Tibayrenc M, Lemesre J, Antezana G, et al. Specific humoral depression in chronic patients infected by Trypanosoma cruzi. Rev Inst Med Trop. 1984;26:254-8. https://doi.org/10.1590/S0036-46651984000500005
Tozetto TR, Vasconcelos DM, Ibrahim KY, Sartori AM, Bezerra RC, Freitas VL, et al. Role of T. cruzi exposure in the pattern of T cell cytokines among chronically infected HIV and Chagas disease patients. Clinics. 2017;72:652-60. https://doi.org/10.6061/clinics/2017(11)02
Taibi A, Guevara A, Schöneck R, Yahiaoui B, Ouaissi A. Improved specificity of Trypanosoma cruzi identification by polymerase chain reaction using an oligonucleotide derived from the amino-terminal sequence of a Tc24 protein. Parasitology. 1995;111:581-90. https://doi.org/10.1017/S0031182000077064
Organización Panamericana de la Salud. Estimación cuantitativa de la enfermedad de Chagas en las Américas. Accessed: April 22, 2019. Available at: http://ops-uruguay.bvsalud.org/pdf/chagas19.pdf
Añez N, Romero M, Crisante G, Bianchi G, Parada H. Valoración comparativa de pruebas serodiagnósticas utilizadas para detectar enfermedad de Chagas en Venezuela. Bol Mal Salud Amb. 2010;50:17-27.
Aria L, Acosta ME, Guillén Y, Rojas A, Meza T, Infanzón B. ELISA Chagas test IICS V. 1 evaluation for the diagnosis of Chagas disease. Mem Inst Investig Cienc Salud. 2016;14:7-13. https://doi.org/10.18004/mem.iics/1812-9528/2016.014(03)07-013
Cermeño JR, Askew EC, Salazar FD. Seroprevalencia de la enfermedad de Chagas en comunidades indígenas de los estados Bolívar y Delta Amacuro, Venezuela. Saber. 2013;25:373-81.
Guzmán-Gómez D, López-Monteón A, De la Soledad Lagunes-Castro M, Álvarez-Martínez C, Hernández-Lutzon MJ, Dumonteil E, et al. Highly discordant serology against Trypanosoma cruzi in central Veracruz, México: Role of the antigen used for diagnostic. Parasit Vectors. 2015;8:466. https://doi.org/10.1186/s13071-015-1072-2
Some similar items:
- Rubén Santiago Nicholls, Zulma Milena Cucunubá, Angélica Knudson, Astrid Carolina Flórez, Marleny Montilla, Concepción Judith Puerta, Paula Ximena Pavía, Acute Chagas disease in Colombia: a rarely suspected disease. Report of 10 cases presented during the 2002-2005 period , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Paula Ximena Pavía, Nubia Lucía Roa, Ana María Uribe, Concepción Judith Puerta, Using S35-S36 and TcH2AF-R primer-based PCR tests to follow-up a Chagas’ disease patient who had undergone a heart transplant , Biomedica: Vol. 31 No. 2 (2011)
- Richard Hoyos, Lisandro Pacheco, Luz Adriana Agudelo, German Zafra, Pedro Blanco, Omar Triana, Seroprevalence of Chagas disease and associated risk factors in a population of Morroa, Sucre , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Concepción Judith Puerta, Johana María Guevara, Paula Ximena Pavía, Marleny Montilla, Rubén Santiago Nicholls, Edgar Parra, Yuli Katherine Barrera, Evaluation of TcH2AF-R and S35-S36 primers in PCR tests for the detection of Trypanosoma cruzi in mouse cardiac tissue , Biomedica: Vol. 28 No. 4 (2008)
- Concepción Judith Puerta, Paula Ximena Pavia, Marleny Montilla, Carolina Flórez, Giomar Herrera, Juan Manuel Ospina, Fred Manrique, Rubén Santiago Nicholls, The first case of congenital Chagas’ disease analyzed by AP-PCR in Colombia , Biomedica: Vol. 29 No. 4 (2009)
- María Clara Echeverry, Nubia Catalina Tovar, Guillermo Mora, Presence of antibodies to cardiac neuroreceptors in patients with Chagas disease , Biomedica: Vol. 29 No. 3 (2009)
- Sandra Paola Santander, Adriana Cuéllar, María del Carmen Thomas, Fanny Guzmán, Alberto Gómez, Manuel Carlos López, Concepción Puerta, Expression of markers on dendritic cells from chronic chagasic patients stimulated with the KMP-11 protein and the K1 peptide from Trypanosoma cruzi , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Juliana Gil, Paula Pavía, Marleny Montilla, Astrid C. Florez, Claudia Quintero, Marcela Mercado, Miguel Vacca, Santiago Nicholls, Concepción Puerta, Comparison of a PCR test based on the histone H2A/SIRE genes with classical serological tests for the diagnosis of chronic Chagas disease in Colombian patients , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Luz Adriana Botero, Ana María Mejía, Omar Triana, Biological and genetic characterization of two Colombian clones of Trypanosoma cruzi groups I and II , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
- Diana Carolina López, Carlos Jaramillo, Felipe Guhl, Population structure and genetic variability of Rhodnius prolixus (Hemiptera: Reduviidae) from different geographic areas of Colombia. , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























