Molecular identification of Entamoeba histolytica, E. dispar, and E. moshkovskii in children with diarrhea from Maracaibo, Venezuela
Abstract
Introduction: Entamoeba histolytica is an amebiasis-producing parasite. However, Entamoeba dispar, Entamoeba moshkovskii, and Entamoeba bangladeshi are nonpathogenic amoebae morphologically identical to it and, therefore, molecular techniques are required for their differentiation.
Objective: To determine the frequency of Entamoeba species by polymerase chain reaction (PCR) in fecal samples from children under five years with diarrhea from Maracaibo (Venezuela).
Materials and methods: A fecal sample per individual was collected from 75 children with diarrhea (case group) and 25 children without diarrhea (control group). Stools were evaluated by microscopic examination, formol-ether concentration method, and nested multiplex PCR in a single round for the identification of E. histolytica, E. dispar, and E. moshkovskii. In addition, a survey was conducted in which demographic data, signs, clinical manifestations, and socioeconomic status were registered.
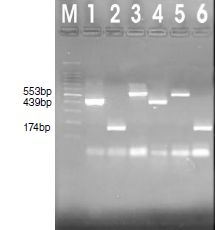
Results: In total, 48% of the children (38 from the case group and 10 from the control group) had intestinal parasites. Only four children presented cysts of the Entamoeba complex in their samples (three from the case group and one from the control group). By means of PCR, nine samples (9%) amplified for the studied amoebae. In the case group, three (28.13%) amplified for E. histolytica, four (30.50%) for E. dispar, and one (9.37%) for E. moshkovskii while only one (25%) sample amplified for E. dispar in the control group.
Conclusion: In general, E. dispar predominated. Nevertheless, all those infected with E. histolytica were detected within the group of children with diarrhea and we reported the first case of E. moshkovskii in the region.
Downloads
References
Ríos-Uil J, Mercadillo-Pérez P, Yuil de Ríos E, Ríos-Castro M. Amebiasis cutánea. Conceptos actuales. Rev Med Hosp Gen Mex. 2012;75:114-22.
Cui Z, Li J, Chen Y, Zhang L. Molecular epidemiology, evolution, and phylogeny of Entamoeba spp. Infec Genet Evol. 2019;75:104018 https://doi.org/10.1016/j.meegid.2019.104018
Carrero J, Reyes-López M, Serrano-Luna J, Shibayama M, Unzueta J, León-Sicairos N, et al. Intestinal amoebiasis: 160 years of its first detection and still remains as a health problem in developing countries. Int J Med Microbiol. 2020;310:151358. https://doi.org/10.1016/j.ijmm.2019.151358
Ngobeni R, Samie A, Moonah S, Watanabe K, Petri WA, Gilchrist C. Entamoeba species in South Africa: Correlations with the host microbiome, parasite burdens, and first description of Entamoeba bangladeshi outside of Asia. J Infect Dis. 2017;216:1592-600. https://doi.org/10.1093/infdis/jix535
Foda AA, El-Malky MM. Prevalence of genital tract infection with Entamoeba gingivalis among copper T 380A intrauterine device users in Egypt. Contraception 2012;85:108-12. https://doi.org/10.1016/j.contraception.2011.04.006
Levecke B, Dorny P, Vercammen F, Visser LG, van Esbroeck M, Vercruysse J, et al. Transmission of Entamoeba nuttalli and Trichuris trichiura from nonhuman primates to humans. Emerg Infect Dis. 2015;21:1871-2. https://doi.org/10.3201/eid2110.141456
Botero D, Restrepo M. Parasitosis humanas. 4ta edición. Medellín: Ediciones Corporación para Investigaciones Biológicas; 2012. p. 725.
Najafia A, Mirzaeia A, Kermanjania A, Abdia J, Ghaderic A, Naserifara R. Molecular identification of Entamoeba histolytica from stool samples of Ilam, Iran. Comp Immunol Microbiol Infect Dis. 2019;63:145-7. https://doi.org/10.1016/j.cimid.2019.01.003
Rivero de Rodríguez Z. Detección de Entamoeba moshkovskii en humanos: un nuevo problema diagnóstico en la amebiasis. Kasmera. 2013;41:42-9.
Gilchrist CA. Entamoeba bangladeshi: An insight. Trop Parasitol. 2014;4:96-8. https://doi.org/10.4103/2229-5070.138536
Quintero R. Crecimiento y desarrollo psicológico al niño venezolano. Puericultura Atención Primaria en Salud infanto-juvenil. Maracaibo: Ediciones Psicopediátricas; 2001. p. 7-10.
Méndez H, De Méndez MC. Sociedad y Estratificación: Método Graffar-Méndez Castellano. Caracas: Editor Fundacredesa; 1994. p. 206.
Melvin D, Brooke M. Métodos de laboratorios para el diagnóstico de parasitosis intestinales. México: Editorial Interamericano; 1971. p. 198.
Rivero Z, Bracho A, Calchi M, Díaz I, Acurero E, Maldonado A, et al. Detección y diferenciación de Entamoeba histolytica y Entamoeba dispar mediante reacción en cadena de la polimerasa en individuos de una comunidad del Estado Zulia, Venezuela. Cad Saúde Pública. 2009;25:151-9. https://doi.org/10.1590/S0102-311X2009000100016
Khairnar K, Parija S. A novel nested multiplex polymerase Chain Reaction (PCR). Assay for differential detection of E. histolytica, E. dispar and moshkovskii DNA in stool samples. BMC Microbiol. 2007;7:47. https://doi.org/10.1186/1471-2180-7-47
Ngui R, Angal L, Fakhrurrazi S, Ai Lian Y, Ling L, Ibrahim J, et al. Differentiating Entamoeba histolytica, Entamoeba dispar and Entamoeba moshkovskii using nested polymerase chain reaction (PCR) in rural communities in Malaysia. Parasit Vectors. 2012;5:187. https://doi.org/10.1186/1756-3305-5-187
Wayne D. Bioestadística. Base para el análisis de las ciencias de la salud. Cuarta edición. México, D.F.: Limusa Wiley; 2002. p. 665.
Bracho A, Rivero de Rodríguez Z, Arraiz N, Villalobos R, Urdaneta H. Detección de Entamoeba histolytica y Entamoeba dispar mediante PCR, en niños menores de cinco años con diarrea, en Maracaibo, Venezuela: estudio preliminar. Invest Clin. 2013;54:373-81.
Samie A, Mahlaule L, Mbati P, Nozaki T, ElBakri A. Prevalence and distribution of Entamoeba species in a rural community in northern South Africa. Food Waterborne Parasitol. 2020;18:e00076. https://doi.org/10.1016/j.fawpar.2020.e00076
Sharbatkhori M, Nazemalhosseini-Mojarad E, Cheraghali F, Maghsoodloorad FS, Taherkhani H, Vakili MA. Discrimination of Entamoeba spp. in children with dysentery. Gastroenterol Hepatol Bed Bench. 2014;7:164-7.
López O, López M, Corredor V, Echeverri C, Pinilla A. Differentiation of Entamoeba histolytica from Entamoeba dispar using Gal/GalNAc-lectin and polymerase chain reaction. Rev Med Chile. 2012;140:476-83. https://doi.org/10.4067/S0034-98872012000400008
Calegar DA, Nunes BC, Monteiro KJ, Santos JP, Toma HK, Gomes TF, et al. Frequency and molecular characterisation of Entamoeba histolytica, Entamoeba dispar, Entamoeba moshkovskii, and Entamoeba hartmanni in the context of water scarcity in northeastern Brazil. Mem Inst Oswaldo Cruz. 2016;111:114-9. https://doi.org/10.1590/0074-02760150383
Abid T. Molecular identification of some species of Entamoeba isolated from patients with diarrhea in Afak city/Qadisiyah governorate using real-time PCR technique. Int J Recent Sci Res. 2016;7:11207-11.
Bahrami F, Haghighi A, Zamini G, Khademerfan M. Differential detection of Entamoeba histolytica, Entamoeba dispar and Entamoeba moshkovskii in faecal samples using nested multiplex PCR in west of Iran. Epidemiol Infect. 2019;147:E96. https://doi.org/10.1017/S0950268819000141
Saidin S, Abu Bakar A, Mohd Zain BM. Prevalence and associated risk factors of Entamoeba histolytica, E. dispar and E. moshkovskii infection among Orang Asli communities in Slim River, Perak. JSML. 2020;8:22-35.
Mora L, García A, De Donato M, Urdaneta H. Estudio epidemiológico y molecular de cepas de Entamoeba histolytica y Entamoeba dispar en pacientes con diarrea en Cumaná, estado Sucre, Venezuela. Invest Clin. 2008;49:225-37.
Roshdy MH, Abd El-Kader NM, Ali-Tammam M, Fuentes I, Mohamed MM, El-Sheikhand NA, et al. Molecular diagnosis of Entamoeba spp. versus microscopy in the Great Cairo. Acta Parasitol. 2017;62:188-91. https://doi.org/10.1515/ap-2017-0022
Haque R, Ali I, Clark C, Petri W. A case report of Entamoeba moshkovskii infection in a Bangladesh child. Parasitol Inter.1998;47:201-2.
Ali IKM, Hossain MB, Roy S, Ayeh-Kumi PF, Petri Jr W, Haque R, et al. Entamoeba moshkovskii infections in children, Bangladesh. Emerg Infect Dis. 2003;9:580-4. https://doi.org/10.3201/eid0905.020548
Levecke B, Dreesen L, Barrionuevo-Samaniego M, Ortiz WB, Praet N, Brandt J, et al. Molecular differentiation of Entamoeba spp. in a rural community of Loja province, South Ecuador. Trans R Soc Trop Med Hyg. 2011;105:737-9. https://doi.org/10.1016/j.trstmh.2011.08.010
Bachkanji AB. Identificación molecular de especies de Entamoeba en muestras fecales provenientes de pacientes del anexo pediátrico del “Hospital Luis Razetti” de Barcelona, Estado Anzoátegui y su relación con síntomas clínicos. (disertación). Anzoátegui: Universidad de Oriente; 2011.
López M, León C, Fonseca J, Reyes P, Moncada L, Olivera M, et al. Molecular epidemiology of Entamoeba: First description of Entamoeba moshkovskii in a rural area from central Colombia. PLoS ONE. 2015;14:1-11 https://doi.org/10.1371/journal.pone.0140302
Soares NM, Azevedo HC, Pacheco FTF, De Souza JN, Del-Rei RP, Teixeira MCA, et al. A cross-sectional study of Entamoeba histolytica/dispar/moshkovskii complex in Salvador, Bahia, Brazil. Biomed Res Int. 2019;2019:7523670. https://doi.org/10.1155/2019/7523670
Ngui R, Hassan N, Soffyan N, Mohd-Shaharuddin N, Chang Li Y, Shuan C, et al. Copromolecular study of Entamoeba infection among the indigenous community in Malaysia: A first report on the species-specific prevalence of Entamoeba in dogs, Acta Tropica. 2020;204:105334. https://doi.org/10.1016/j.actatropica.2020.105334
Castro AA, Bacalhau F, Silva FF, Avillez C, Batalheiro J. Entamoeba histolytica como causa de diarreia crônica. Rev Bras Med Fam Comunidade. 2019;14:1917. https://doi.org/10.5712/rbmfc14(41)1917
Aguilar-Solís BE, Sánchez-Rodríguez A, Sael-Lima M, Álvarez-Trejo VE. Amebiasis vulvar. Dermatol Rev Mex. 2017;61:142-6.
Blessmann J, Ali IK, Nu PA, Dinh BT, Viet TQ, Van AL, et al. Longitudinal study of intestinal Entamoeba histolytica infections in asymptomatic adult carriers. J Clin Microbiol. 2003;41:4745-50. https://doi.org/10.1128/jcm.41.10.4745-4750.2003
Andrade A, Santana T. Entamoeba histolytica como causa da amebíase. Rev Saúde Meio Ambiente. 2020;10:133-9
Aguilar-Rojas A, Castellanos-Castro S, Matondo M, Giai Q, Varet H, Sismeiro O, et al. Insights into amebiasis using a human 3D-intestinal model. Cell Microbiol. 2020;22:e13203. https://doi.org/10.1111/cmi.13203
Hernández C, Moreno J, Olarte M, Meza E, Regalado J. Amebiasis intestinal: infección que prevalece. Ibn Sina. 2020;11:1-11.
Saavedra E, Olivos A. Amebiasis. Ciencia. 2017;68:14-7.
Ugboko HU, Nwinyi OC, Oranusi SU, Oyewal JO. Childhood diarrhoeal diseases in developing countries. Heliyon. 2020;6:e03690 https://doi.org/10.1016/j.heliyon.2020.e03690
Haque R, Mondai D, Kirkpatrick B, Akter S, Farr B, Sack B, et al. Epidemiologic and clinical characteristics of acute diarrhea with emphasis on Entamoeba histolytica infections in preschool children in an urban slum of Dhaka, Bangladesh. Am J Trop Med Hyg. 2003;69:398-405.
Akisu C, Aksoy U, Cetin H, Ustun S, Akisu M. Effect of human milk and colostrum on Entamoeba histolytica. World J Gastroenterol. 2004;10:741-2. https://doi.org/10.3748/wjg.v10.i5.741
Devera R, Aguilar K, Maurera R, Blanco Y, Amaya I, Velásquez V. Parasitosis intestinales en alumnos de la escuela básica nacional: “San José de Cacahual”. San Félix, Estado Bolívar, Venezuela. Rev Academia. 2016;15:35-46.
Cociancica P, Torrusio S, Zonta ML, Navone G. Risk factors for intestinal parasitoses among children and youth of Buenos Aires, Argentina. One Health. 2020;9:100116. https://doi.org/10.1016/j.onehlt.2019.100116
Gholipoor Z, Khazan H, Azargashb E, Youssefi M, Rostami A. Prevalence and risk factors of intestinal parasite infections in Mazandaran province, North of Iran. Clin Epidemiol Global Health 2020;8:17-20. https://doi.org/10.1016/j.cegh.2019.03.010
Some similar items:
- Nelsy Loango, Martha Lucía Gallego, Beatriz Restrepo, Patricia Landázuri, Gender, age and plasma lipids differences associated with apolipoprotein E polymorphism in school children , Biomedica: Vol. 29 No. 3 (2009)
- Rosa Magdalena Uscátegui, Adriana M. Correa, Jaime Carmona-Fonseca, Changes in retinol, hemoglobin and ferritin concentrations in Colombian children with malaria , Biomedica: Vol. 29 No. 2 (2009)
- Andrés Leonardo González, Ruth Aralí Martínez, Luis Ángel Villar, Clinical evolution of dengue in hospitalized patients , Biomedica: Vol. 28 No. 4 (2008)
- Elsa Nieves, Néstor Villarreal, Maritza Rondón, Mireya Sánchez, José Carrero, Evaluation of knowledge and practice on tegumentary leishmaniasis in an endemic area of Venezuela , Biomedica: Vol. 28 No. 3 (2008)
- Elpidia Poveda, Diana Giraldo, Yibby Forero, Carlos Mendivil, Self-reported physical activity in comparison with anthropometric body fat indicators in school children , Biomedica: Vol. 28 No. 3 (2008)
- Ligia Inés Moncada, Sandra Milena Rios, Julián Alfredo Fernández, Fabio Rivas, María Luz Sáenz, Pediculosis prevalence and associated risk factors in a nursery school, Bogotá, Colombia , Biomedica: Vol. 28 No. 2 (2008)
- Elsa Villarreal, Yibby Forero, Elpidia Poveda, César Baracaldo, Elizabeth López, Cardiovascular risk markers in schoolchildren from five provinces of eastern Colombia , Biomedica: Vol. 28 No. 1 (2008)
- Olga Serrano, Florencio Mendoza, Benny Suárez, Ana Soto, Seroepidemiology of Chagas disease in two rural populations in the municipality of Costa de Oro, at Aragua State, northern Venezuela , Biomedica: Vol. 28 No. 1 (2008)
- Jairo Echeverry, Carlos Mauricio Hurtado, Myriam Gutiérrez, Clinical manifestations of lead levels in children exposed to automobile battery recycling processes in Soacha and Bogotá, D.C. , Biomedica: Vol. 28 No. 1 (2008)
- Elpidia Poveda, Ney E. Callas, César M. Baracaldo, Carlina Castillo, Patricia Hernández, Leptin levels in school age children associated with anthropometric measurements and lipid profiles , Biomedica: Vol. 27 No. 4 (2007)
Funding data
-
Consejo de Desarrollo Científico, Humanístico y Tecnológico, Universidad del Zulia
Grant numbers CONDES-0490-13
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























