Los aptámeros como novedosa herramienta diagnóstica y terapéutica y su potencial uso en parasitología
Resumen
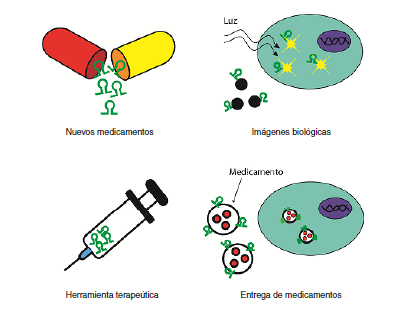
Los aptámeros son secuencias de ADN o ARN de cadena sencilla que adoptan la forma de estructuras tridimensionales únicas, lo cual les permite reconocer un blanco específico con gran afinidad. Sus usos potenciales abarcan, entre otros, el diagnóstico de enfermedades, el desarrollo de nuevos agentes terapéuticos, la detección de riesgos alimentarios, la producción de biosensores, la detección de toxinas, el transporte de fármacos en el organismo y la señalización de nanopartículas.
El pegaptanib es el único aptámero aprobado para uso comercial por la Food and Drug Administration (FDA).
En parasitología, se destacan los estudios que se vienen realizando en Leishmania spp., con la obtención de aptámeros que reconocen la proteína de unión a poliA (LiPABP) y que pueden tener potencial utilidad en la investigación, el diagnóstico y el tratamiento de la leishmaniasis. En cuanto a la malaria, se han obtenido aptámeros que permiten identificar eritrocitos infectados e inhiben la formación de rosetas, y otros que prometen ser alternativas para el diagnóstico al detectar de forma específica la proteína lactato deshidrogenasa (PfLDH). Para Cryptosporidium parvuum se han seleccionado aptámeros que detectan ooquistes a partir de alimentos o aguas contaminadas. Para Entamoeba histolytica se han aislado dos aptámeros llamados C4 y C5, que inhiben la proliferación in vitro de los trofozoítos y tienen potencial terapéutico. Los aptámeros contra Trypanosoma cruzi inhiben la invasión de células LLC-MK2 (de riñón de mono) en un 50 a 70 % y aquellos contra T. brucei transportan moléculas tóxicas al lisosoma parasitario como una novedosa estrategia terapéutica.
Descargas
Referencias bibliográficas
Shannon-Pendergrast P, Nicholas-Marsh H, Grate D, Healy JM, Stanton M. Nucleic acid aptamers for target validation and therapeutic applications. J Biomol Tech. 2005;16:224-34.
Shigdar S, Qian C, Lv L, Pu C, Li Y, Li L, et al. The use of sensitive chemical antibodies for diagnosis: Detection of low levels of Epcam in breast cancer. PLoS One. 2013;8:e57613. https://doi.org/10.1371/journal.pone.0057613
Ellington AD, Szostak JW. In vitro selection of RNA molecules that bind specific ligands. Nature. 1990;346:818-22. https://doi.org/10.1038/346818a0
Tuerk C, Gold L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science. 1990;249:505-10. https://doi.org/10.1126/science.2200121
Catherine AT, Shishido SN, Robbins-Welty GA, Diegelman-Parente A. Rational design of a structure-switching DNA aptamer for potassium ions. FEBS Open Bio. 2014;4:788-95. https://doi.org/10.1016/j.fob.2014.08.008
Kratschmer C, Levy M. Targeted delivery of auristatin-modified toxins to pancreatic cancer using aptamers. Mol Ther Nucleic Acids. 2018;10:227-36. https://doi.org/10.1016/j.omtn.2017.11.013
Heilkenbrinker A, Reinemann C, Stoltenburg R, Walter JG, Jochums A, Stahl F, et al. Identification of the target binding site of ethanolamine-binding aptamers and its exploitation for ethanolamine detection. Anal Chem. 2015;87:677-85. https://doi.org/10.1021/ac5034819
Xing YP, Liu C, Zhou XH, Shi HC. Label-free detection of kanamycin based on a G-quadruplex DNA aptamer-based fluorescent intercalator displacement assay. Sci Rep. 2015;5:14-6. https://doi.org/10.1038/srep08125
Geiger A, Burgstaller P, von der Eltz H, Roeder A, Famulok M. RNA aptamers that bind L-arginine with sub-micromolar dissociation constants and high enantioselectivity. Nucleic Acids Res. 1996;24:1029-36. https://doi.org/10.1093/nar/24.6.1029
Xu W, Ellington AD. Anti-peptide aptamers recognize amino acid sequence and bind a protein epitope. Proc Natl Acad Sci USA. 1996;93:7475-80. https://doi.org/10.1073/pnas.93.15.7475
Yang Q, Goldstein IJ, Mei H-Y, Engelke DR. DNA ligands that bind tightly and selectively to cellobiose. Proc Natl Acad Sci.1998;95:5462-7. https://doi.org/10.1073/pnas.95.10.5462
Ospina-Villa JD, Dufour A, Weber C, Ramírez-Moreno E, Zamorano-Carrillo A, Guillen N. Targeting the polyadenylation factor EhCFIm25 with RNA aptamers controls survival in Entamoeba histolytica. Sci Rep. 2018;8:5720. https://doi.org/10.1038/s41598-018-23997-w
Trujillo CA, Nery AA, Alves JM, Martins AH, Ulrich H. Development of the anti-VEGF aptamer to a therapeutic agent for clinical ophthalmology. Clin Ophthalmol. 2007;1:393-402.
Percze K, Szakács Z, Scholz É, András J, Szeitner Z, Kieboom CH, et al. Aptamers for respiratory syncytial virus detection. Sci Rep. 2017;7:1-11. https://doi.org/10.1038/srep42794
Song MY, Nguyen D, Hong SW, Kim BC. Broadly reactive aptamers targeting bacteria belonging to different genera using a sequential toggle cell-SELEX. Sci Rep. 2017;7:43641. https://doi.org/10.1038/srep43641
Nagarkatti R, Bist V, Sun S, Fortes de Araujo F, Nakhasi HL, Debrabant A. Development of an aptamer-based concentration method for the detection of Trypanosoma cruzi in blood. PLoS One. 2012;7:e43533. https://doi.org/10.1371/journal.pone.0043533
Zhou J, Rossi JJ. Cell-type-specific, aptamer-functionalized agents for targeted disease therapy. Mol Ther Nucleic Acids. 2014;3:e169. https://doi.org/10.1038/mtna.2014.21
Richards SL, Cawley AT, Cavicchioli R, Suann CJ, Pickford R, Raftery MJ. Aptamer based peptide enrichment for quantitative analysis of gonadotropin-releasing hormone by LC-MS/MS. Talanta. 2016;150:671-80. https://doi.org/10.1016/j.talanta.2016.01.006
Zhan S, Yu M, Lv J, Wang L, Zhou P. Colorimetric detection of trace arsenic (III) in aqueous solution using arsenic aptamer and gold nanoparticles. Aust J Chem. 2014;67:813-8.
Sachan A, Ilgu M, Kempema A, Kraus GA, Nilsen-Hamilton M. Specificity and ligand affinities of the cocaine aptamer: Impact of structural features and physiological NaCl. Anal Chem. 2016;88:7715-23. https://doi.org/10.1021/acs.analchem.6b01633
Park KS. Nucleic acid aptamer-based methods for diagnosis of infections. Biosens Bioelectron. 2018;102:179-88. https://doi.org/10.1016/j.bios.2017.11.028
Wandtke T, Woźniak J, Kopiński P. Aptamers in diagnostics and treatment of viral infections. Viruses. 2015;7:751-80. https://doi.org/10.3390/v7020751
Zhao N, Pei SN, Qi J, Zeng Z, Iyer SP, Lin P, et al. Oligonucleotide aptamer-drug conjugates for targeted therapy of acute myeloid leukemia. Biomaterials. 2015;67:42-51. https://doi.org/10.1016/j.biomaterials.2015.07.025
Tan W, Donovan MJ, Jiang J. Aptamers from cell-based selection for bioanalytical applications. Chem Rev. 2013;113:2842-62. https://doi.org/10.1021/cr300468w
Song S, Wang L, Li J, Fan C, Zhao J. Aptamer-based biosensors. TrAC - Trends Anal Chem. 2008;27:108-17. https://doi.org/10.1016/j.trac.2007.12.004
Dong Y, Xu Y, Yong W, Chu X, Wang D. Aptamer and its potential applications for food safety. Crit Rev Food Sci Nutr. 2014;54:1548-61. https://doi.org/10.1080/10408398.2011.642905
Liu J, Wei T, Zhao J, Huang Y, Deng H, Kumar A, et al. Multifunctional aptamer-based nanoparticles for targeted drug delivery to circumvent cancer resistance. Biomaterials. 2016;91:44-56. https://doi.org/10.1016/j.biomaterials.2016.03.013
Yoon S, Rossi JJ. Future strategies for the discovery of therapeutic aptamers. Expert Opin Drug Discov. 2017;12:317-9. https://doi.org/10.1080/17460441.2017.1290077
Gold L, Ringquist S. Systematic evolution of ligands by exponential enrichment: Solution SELEX. Fecha de consulta: 20 de septiembre de 2019. Disponible en: https://patents.google.com/patent/US5567588A/en
MarketsandMarkets. Aptamers Market Size & Share by Application (Diagnostics, Therapeutics, R&D), Type (DNA, RNA, XNA), Technology (SELEX, Other Technologies) & End Users (Academic Research Institutes, Biotechnology & Pharmaceutical Companies, CRO) - Global Forecast to 2020. Fecha de consulta: 10 de septiembre de 2019. Disponible en: https://www.marketsandmarkets.com/Market-Reports/aptamers-technology-market-1167.html
Katz M. Adverse metabolic effects of antiparasitic drugs. Rev Infect Dis. 1982;4:768-70.
Kang D, Wang J, Zhang W, Song Y, Li X, Zou Y, et al. Selection of DNA aptamers against glioblastoma cells with high affinity and specificity. PLoS One. 2012;7:e42731. https://doi.org/10.1371/journal.pone.0042731
Liu M, Wang Z, Tan T, Chen Z, Mou X, Yu X, et al. An aptamer-based probe for molecular subtyping of breast cancer. Theranostics. 2018 8:5772-83. https://doi.org/10.7150/thno.28949
Chen L, Rashid F, Shah A, Awan HM, Wu M, Liu A, et al. The isolation of an RNA aptamer targeting to p53 protein with single amino acid mutation. Proc Natl Acad Sci USA. 2015;112:10002-7. https://doi.org/10.1073/pnas.1502159112
Zhang H, Li XF, Le XC. Differentiation and detection of PDGF isomers and their receptors by tunable aptamer capillary electrophoresis. Anal Chem. 2009;81:7795-800. https://doi.org/10.1021/ac901471w
Mendonsa SD, Bowser MT. In vitro evolution of functional DNA using capillary electrophoresis. J Am Chem Soc. 2004;126:20-1. https://doi.org/10.1021/ja037832s
Krylov SN. Nonequilibrium capillary electrophoresis of equilibrium mixtures (NECEEM): A novel method for biomolecular screening. J Biomol Screen. 2006;11:115-22. https://doi.org/10.1177/1087057105284339
Hybarger G, Bynum J, Williams RF, Valdes JJ, Chambers JP. A microfluidic SELEX prototype. Anal Bioanal Chem. 2006;384:191-8. https://doi.org/10.1007/s00216-005-0089-3
Ogawa N, Biggin MD. High-throughput SELEX determination of DNA sequences bound by ranscription factors in vitro. Methods Mol Biol. 2012;786:51-63. https://doi.org/10.1007/978-1-61779-292-2_3
Kong HY, Byun J. Nucleic acid aptamers: New methods for selection, stabilization, and application in biomedical science. Biomol Ther (Seoul). 2013;21:423-34. https://doi.org/10.4062/biomolther.2013.085
Zhuo Z, Yu Y, Wang M, Li J, Zhang Z, Liu J, et al. Recent advances in SELEX technology and aptamer applications in biomedicine. Int J Mol Sci. 2017;18:1-19. https://doi.org/10.3390/ijms18102142
Ni S, Yao H, Wang L, Lu J, Jiang F, Lu A, et al. Chemical modifications of nucleic acid aptamers for therapeutic purposes. Int J Mol Sci. 2017;18. https://doi.org/10.3390/ijms18081683
Haruta K, Otaki N, Nagamine M, Kayo T, Sasaki A, Hiramoto S, et al. A novel PEGylation method for improving the pharmacokinetic properties of anti-interleukin-17A RNA aptamers. Nucleic Acid Ther. 2017;27:36-44. https://doi.org/10.1089/nat.2016.062
Maier KE, Levy M. From selection hits to clinical leads: Progress in aptamer discovery. Mol Ther Methods Clin Dev. 2016;3:16014. https://doi.org/10.1038/mtm.2016.14
Darfeuille F, Hansen JB, Orum H, Di Primo C, Toulmé JJ. LNA/DNA chimeric oligomers mimic RNA aptamers targeted to the TAR RNA element of HIV-1. Nucleic Acids Res. 2004;32:3101-7. https://doi.org/10.1093/nar/gkh636
Nomura Y, Sugiyama S, Sakamoto T, Miyakawa S, Adachi H, Takano K, et al. Conformational plasticity of RNA for target recognition as revealed by the 2.15 Å crystal structure of a human IgG-aptamer complex. Nucleic Acids Res. 2010;38:7822-9. https://doi.org/10.1093/nar/gkq615
Long SB, Long MB, White RR, Sullenger BA. Crystal structure of an RNA aptamer bound to thrombin. RNA. 2008;14:2504-12. https://doi.org/10.1261/rna.1239308
Ruigrok VJB, Levisson M, Hekelaar J, Smidt H, Dijkstra BW, van der Oost J. Characterization of aptamer-protein complexes by x-ray crystallography and alternative approaches. Int J Mol Sci. 2012;13:10537-52. https://doi.org/10.3390/ijms130810537
Sakamoto T, Ennifar E, Nakamura Y. Thermodynamic study of aptamers binding to their target proteins. Biochimie. 2018;145:91-7. https://doi.org/10.1016/j.biochi.2017.10.010
Amano R, Takada K, Tanaka Y, Nakamura Y, Kawai G, Kozu T, et al. Kinetic and thermodynamic analyses of interaction between a high-affinity RNA aptamer and its target protein. Biochemistry. 2016;55:6221-9. https://doi.org/10.1021/acs.biochem.6b00748
Hermann T, Patel DJ. Adaptive recognition by aptamers nucleic acid. Science. 2000;287:820-5. https://doi.org/10.1126/science.287.5454.820
Lin PH, Tsai CW, Wu JW, Ruaan RC, Chen WY. Molecular dynamics simulation of the induced-fit binding process of DNA aptamer and L-argininamide. Biotechnol J. 2012;7:1367-75. https://doi.org/10.1002/biot.201200003
Munzar JD, Ng A, Juncker D. Comprehensive profiling of the ligand binding landscapes of duplexed aptamer families reveals widespread induced fit. Nat Commun. 2018;9:343. https://doi.org/10.1038/s41467-017-02556-3
Serganov A, Nudler E. A decade of riboswitches. Cell. 2013;152:17-24. https://doi.org/10.1016/j.cell.2012.12.024
Nguyen GT, Scaife MA, Helliwell KE, Smith AG. Role of riboswitches in gene regulation and their potential for algal biotechnology. J Phycol. 2016;52:320-8. https://doi.org/10.1111/jpy.12416
Gupta A, Swati D. Exploring riboswitches in archaeal metagenomes. J RNAi Gene Silenc. 2016;12:536-43.
Cheah MT, Wachter A, Sudarsan N, Breaker RR. Control of alternative RNA splicing and gene expression by eukaryotic riboswitches. Nature. 2007;447:497-500. https://doi.org/10.1038/nature05769
Ng EW, Shima DT, Calias P, Cunningham Jr ET, Guyer DR, Adamis AP. Pegaptanib, a targeted anti-VEGF aptamer for ocular vascular disease. Nat Rev Drug Discov. 2006;5:123. https://doi.org/10.1038/nrd1955
Vinores SA. Pegaptanib in the treatment of wet, age-related macular degeneration. Int J Nanomedicine. 2006;1:263-8.
Chen Q. The naturally acquired immunity in severe malaria and its implication for a PfEMP-1 based vaccine. Microbes Infect. 2007;9:777-83. https://doi.org/10.1016/j.micinf.2007.02.009
Barfod A, Persson T, Lindh J. In vitro selection of RNA aptamers against a conserved region of the Plasmodium falciparum erythrocyte membrane protein 1. Parasitol Res. 2009;105:1557-66. https://doi.org/10.1007/s00436-009-1583-x
Normark J, Nilsson D, Ribacke U, Winter G, Moll K, Wheelock CE, et al. PfEMP1-DBL1alpha amino acid motifs in severe disease states of Plasmodium falciparum malaria. Proc Natl Acad Sci USA. 2007;104:15835-40. https://doi.org/10.1073/pnas.0610485104
Birch CM, Hou HW, Han J, Niles JC. Identification of malaria parasite-infected red blood cell surface aptamers by inertial microfluidic SELEX (I-SELEX). Sci Rep. 2015;5:11347. https://doi.org/10.1038/srep11347
Cheung Y-W, Kwok J, Law AW, Watt RM, Kotaka M, Tanner JA. Structural basis for discriminatory recognition of Plasmodium lactate dehydrogenase by a DNA aptamer. Proc Natl Acad Sci USA. 2013;110:15967-72. https://doi.org/10.1073/pnas.1309538110
Cheung YW, Dirkzwager RM, Wong WC, Cardoso J, D’Arc Neves Costa J, Tanner JA. Aptamer-mediated Plasmodium-specific diagnosis of malaria. Biochimie. 2018;145:131-6. https://doi.org/10.1016/j.biochi.2017.10.017
Fraser LA, Kinghorn AB, Dirkzwager RM, Liang S, Cheung YW, Lim B, et al. A portable microfluidic Aptamer-Tethered Enzyme Capture (APTEC) biosensor for malaria diagnosis. Biosens Bioelectron. 2018;100:591-6. https://doi.org/10.1016/j.bios.2017.10.001
Niles JC, Derisi JL, Marletta MA. Inhibiting Plasmodium falciparum growth and heme detoxification pathway using heme-binding DNA aptamers. Proc Natl Acad Sci USA. 2009;106:13266-71. https://doi.org/10.1073/pnas.0906370106
Brown KM, Gilmartin GM. A mechanism for the regulation of pre-mRNA 3’ processing by human cleavage factor Im. Mol Cell. 2003;12:1467-76. https://doi.org/10.1016/S1097-2765(03)00453-2
Ospina-Villa JD, Guillén N, López-Camarillo C, Soto-Sánchez J, Ramírez-Moreno E, García-Vázquez R, et al. Silencing the cleavage factor CFIm25 as a new strategy to control Entamoeba histolytica parasite. J Microbiol. 2017;55:783-91. https://doi.org/10.1007/s12275-017-7259-9
Ramos E, Pĩeiro D, Soto M, Abanades DR, Martín ME, Salinas M, et al. A DNA aptamer population specifically detects Leishmania infantum H2A antigen. Lab Investig. 2007;87:409-16. https://doi.org/10.1038/labinvest.3700535
Martín ME, García-Hernández M, García-Recio EM, Gómez-Chacón GF, Sánchez-López M, González VM. DNA aptamers selectively target Leishmania infantum H2A protein. PLoS One. 2013;8:1-11. https://doi.org/10.1371/journal.pone.007888
Guerra-Pérez N, Ramos E, García-Hernández M, Pinto C, Soto M, Martín ME, et al. Molecular and functional characterization of ssDNA aptamers that specifically bind Leishmania infantum PABP. PLoS One. 2015;10:1-17. https://doi.org/10.1371/journal.pone.0140048
Iqbal A, Labib M, Muharemagic D, Sattar S, Dixon BR, Berezovski MV. Detection of Cryptosporidium parvum oocysts on fresh produce using DNA aptamers. PLoS One. 2015;10:1-13. https://doi.org/10.1371/journal.pone.0137455
Ulrich H, Magdesian MH, Alves MJM, Colli W. In vitro selection of RNA aptamers that bind to cell adhesion receptors of Trypanosoma cruzi and inhibit cell invasion. J Biol Chem. 2002;277:20756-62. https://doi.org/10.1074/jbc.M111859200
Nagarkatti R, de Araujo FF, Gupta C, Debrabant A. Aptamer based, non-PCR, non-serological detection of Chagas disease biomarkers in Trypanosoma cruzi infected mice. Burleigh BA, editor. PLoS Negl Trop Dis. 2014;8:e2650. https://doi.org/10.1371/journal.pntd.0002650
Homann M, Göringer HU. Uptake and intracellular transport of RNA aptamers in African trypanosomes suggest therapeutic “piggy-back” approach. Biorg Med Chem. 2001;9:2571-80. https://doi.org/10.1016/S0968-0896(01)00032-3
Gold L. SELEX: How it happened and where it will go. J Mol Evol. 2015;81:140-3. https://doi.org/10.1007/s00239-015-9705-9
Algunos artículos similares:
- Elizabeth Borrero, Gabriel Carrasquilla, Neal Alexander, Descentralización y reforma: ¿cuál es su impacto sobre la incidencia de malaria en los municipios colombianos? , Biomédica: Vol. 32 (2012): Suplemento 1, Malaria
- Iveth J. González, Las metacaspasas y su rol en la vida y muerte de los parásitos protozoarios humanos , Biomédica: Vol. 29 Núm. 3 (2009)
- Rosa Magdalena Uscátegui, Adriana M. Correa, Jaime Carmona-Fonseca, Cambios en las concentraciones de retinol, hemoglobina y ferritina en niños palúdicos colombianos , Biomédica: Vol. 29 Núm. 2 (2009)
- Ana María Vásquez, Felipe Sanín, Luis Gonzalo Álvarez, Alberto Tobón, Alexandra Ríos, Silvia Blair, Estudio piloto de la eficacia y de los efectos sobre los gametocitos del esquema artesunato-mefloquina-primaquina para la malaria por Plasmodium falciparum , Biomédica: Vol. 29 Núm. 2 (2009)
- Alberto Tobón, Signos de peligro en el paciente con malaria , Biomédica: Vol. 29 Núm. 2 (2009)
- Amanda Maestre, Jaime Carmona-Fonseca, Amanda Maestre, Alta frecuencia de mutaciones puntuales en pfcrt de Plasmodium falciparum y emergencia de nuevos haplotipos mutantes en Colombia , Biomédica: Vol. 28 Núm. 4 (2008)
- Nicolás Jaramillo, David Alonso Calle, Martha Lucía Quiñones, Holmes Francisco Erazo, Discriminación por morfometría geométrica de once especies de Anopheles (Nyssorhynchus) presentes en Colombia , Biomédica: Vol. 28 Núm. 3 (2008)
- Angélica Knudson, Rubén Santiago Nicholls, Ángela Patricia Guerra, Ricardo Sánchez, Perfiles clínicos del paludismo no complicado por Plasmodium falciparum en Córdoba, Colombia , Biomédica: Vol. 27 Núm. 4 (2007)
- Rafael José Vivero, Maria Angélica Contreras-Gutiérrez, Eduar Elías Bejarano, Análisis de la estructura primaria y secundaria del ARN de transferencia mitocondrial para serina en siete especies de Lutzomyia , Biomédica: Vol. 27 Núm. 3 (2007)
- Paula Montoya, Alberto Tobón, Silvia Blair, Jaime Carmona, Amanda Maestre, Polimorfismos del gen pfmdr1 en muestras clínicas de Plasmodium falciparum y su relación con la respuesta terapéutica a antipalúdicos y paludismo grave en Colombia , Biomédica: Vol. 27 Núm. 2 (2007)
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























