Shedding of HSV-1, HSV-2, CMV, and EBV in the saliva of hematopoietic stem cell transplant recipients at Fundación HOMI - Hospital de la Misericordia, Bogotá, D.C.
Abstract
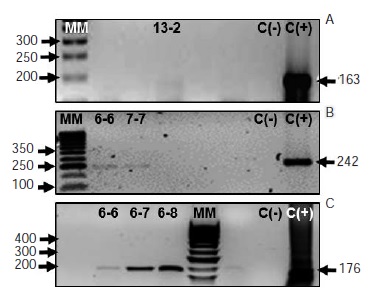
Introduction: Hematopoietic stem cell transplantation in pediatric patients is an alternative treatment for different diseases. The conditioning regimen for transplant predisposes recipients to the development of infections. Viral infections by herpes simplex virus 1 (HSV-1), herpes simplex virus 2 (HSV-2), human cytomegalovirus (CMV), and Epstein-Barr virus (EBV), are the most common, and the leading cause of morbidity and mortality among these patients. These viruses lie dormant in various cell types and the reactivation of latent infections may lead to asymptomatic viral shedding in saliva. The detection of these viruses in secretions may contribute to understand the behavioral dynamics of these viral infections in transplanted patients, and to the early diagnosis of reactivation. Objective: To assess HSV-1, HSV-2, CMV and EBV viral shedding in the saliva of patients admitted for hematopoietic stem cell transplantation at Fundación HOMI - Hospital de la Misericordia between January and November of 2012. Materials and methods: We evaluated stimulated saliva samples of 17 hematopoietic stem cell transplantation recipients weekly. We performed DNA extraction from saliva, and we evaluated the presence of DNA for HSV-1, HSV-2, CMV, and EBV by PCR. Results: While we detected HSV-2 and CMV DNA in the saliva of four patients, EBV DNA was detected in nine patients with leukopenia. In contrast, we did not detect HSV-1 DNA in saliva. Additionally, four out of the 17 patients showed a simultaneous shedding of CMV and EBV. Conclusions: By conventional PCR, we demonstrated asymptomatic HSV-2, CMV, and EBV viral shedding in saliva, associated with leukopenia.
Downloads
References
Whitley RJ, Roizman B. Herpes simplex virus infections. Lancet. 2001;357:1513-8. http://dx.doi.org/10.1016/S0140-6736(00)04638-9
Doumas S, Vladikas A, Papagianni M. Kolokotronis A. Human cytomegalovirus-associated oral and maxillo-facial disease. Clin Microbiol Infect. 2007;13:557-9. http://dx.doi.org/10.1111/j.1469-0691.2007.01714.x
Davison AJ. Herpesvirus systematics. Vet Microbiol. 2010; 143:52-69. http://dx.doi.org/10.1016/j.vetmic.2010.02.014
Wald A, Corey L. Persistence in the population: Epidemiology and transmission. En: Arvin A, Campadelli-Fiume G, Mocarski E, Moore PS, Roizman B, Whitley R, et al., editors. Human herpesviruses: Biology, therapy and immunoprophylaxis. Cambridge: Cambridge University Press;2007. p. 1-31.
Wilson AC, Mohr I. A cultured affair: HSV latency and reactivation in neurons. Trends Microbiol. 2012;20:604-11. http://dx.doi.org/10.1016/j.tim.2012.08.005
Sinclair JH, Reeves MB. Human cytomegalovirus manipu-lation of latently infected cells. Viruses. 2013;5:2803-24. http://dx.doi.org/doi:10.3390/v5112803
Tsurumi T, Fujita M, Kudoh A. Latent and lytic Epstein-Barr virus replication strategies. Rev Med Virol. 2005;15:3-15. http://dx.doi.org/10.1002/rmv.441
Sacks SL, Griffiths PD, Corey L, Cohen C, Cunningham A, Dusheiko GM, et al. HSV shedding. Antiviral Res. 2004;63(Supl.1):S19-26. http://dx.doi.org/10.1016/j.antiviral. 2004.06.004
Sepúlveda E, Rojas IG, Brethauer U, Maulén NP, Muñoz M, Kirsten L, et al. Effect of white cell counts on the presence of human herpes simplex virus type-1 in saliva of pediatric oncology patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105:583-8. http://dx.doi.org/10.1016/j.tripleo.2007.12.035
Wald A, Eriksson M, Krantz E, Selke S, Corey L. Oral shedding of herpes simplex virus type 2. Sex Transm Infect. 2004;80:272-6. http://dx.doi.org/10.1136/sti.2003.007823
Morfin F, Bilger K, Boucher A, Thiebaut A, Najioullah F, Bleyzac N, et al. HSV excretion after bone marrow transplantation: A 4-year survey. J Clin Virol. 2004;30:341-5. http://dx.doi.org/10.1016/j.jcv.2004.03.002
Cannon MJ, Hyde TB, Schmid DS. Review of cytomega-lovirus shedding in bodily fluids and relevance to congenital cytomegalovirus infection. Rev Med Virol. 2011;21:240-55. http://dx.doi.org/10.1002/rmv.695
Jaskula E, Dlubek D, Sedzimirska M, Duda D, Tarnowska A, Lange A. Reactivations of cytomegalovirus, human herpes virus 6, and Epstein-Barr virus differ with respect to risk factors and clinical outcome after hematopoietic stem cell transplantation. Transplant Proc. 2010;42:3273-6. http://dx.doi.org/10.1016/j.transproceed.2010.07.027
Hug M, Dorner M, Fröhlich FZ, Gysin C, Neuhaus D, Nadal D. Pediatric Epstein-Barr virus carriers with or without tonsillar enlargement may substantially contribute to spreading of the virus. J Infect Dis. 2010;202:1192-9. http://dx.doi.org/10.1086/656335
Lucht E, Biberfeld P, Linde A. Epstein-Barr virus (EBV) DNA in saliva and EBV serology of HIV-1-infected persons with and without hairy leukoplakia. J Infect. 1995;31:189-94.
Cordonnier C. Infections after HSCT. In: Apperley J, Carreras E, Gluckman E, Gratwohl A, Masszi T, editors. ESH-EBMT Handbook. Haematopoietic Stem Cell Transplantation. 5th edition. Paris: European School of Haematology; 2008. p. 198-217.
Correia-Silva JdeF, Victória JM, Guimarães AL, Salomão UE, de Abreu MH, Bittencourt H, et al. Cytomegalovirus shedding in the oral cavity of allogeneic haematopoietic stem cell transplant patients. Oral Dis. 2007;13:163-9. http://dx.doi.org/10.1111/j.1601-0825.2006.01240.x
Preiksaitis JK, Díaz-Mitoma F, Mirzayans F, Roberts S, Tyrrell DL. Quantitative oropharyngeal Epstein-Barr virus shedding in renal and cardiac transplant recipients: Relation-ship to immunosuppressive therapy, serologic responses, and the risk of post-transplant lymphoproliferative disorder. J Infect Dis.1992;166:986-94. http://dx.doi.org/10.1093/infdis/166.5.986
Perera RA, Samaranayake LP, Tsang CS. Shedding dynamics of Epstein-Barr virus: A type 1 carcinogen. Arch Oral Biol. 2010;55:639-47. http://dx.doi.org/10.1016/j.archoralbio.2010.06.009
Kessler HH, Pierer K, Weber B, Sakrauski A. Detection of herpes simplex virus DNA from cerebrospinal fluid by PCR and a rapid, nonradioactive hybridization technique. J Clin Microbiol. 1994;32:1881-6.
Sun Y, Kum R, Hoon S, Pei P. Detection and genotyping of human herpes simplex viruses in cutaneous lesions of erythema multiforme by nested PCR. J Med Virol. 2003;71: 423-8. http://dx.doi.org/10.1002/jmv.10502
Shin CH, Park GS, Hong KM, Paik MK. Detection and typing of HSV-1, HSV-2, CMV and EBV by quadruplex PCR. Yonsei Med J. 2003;44:1001-7. http://dx.doi.org/10.3349/ymj.2003.44.6.1001
Sahin F, Gerceker D, Karasartova D, Ozsan TM. Detection of herpes simplex virus type 1 in addition to Epstein-Bar virus in tonsils using a new multiplex polymerase chain reaction assay. Diagn Microbiol Infect Dis. 2007;57:47-51. http://dx. doi.org/10.1016/j.diagmicrobio.2006.09.013
Miller CS, Danaher RJ. Asymptomatic shedding of herpes simplex virus (HSV) in the oral cavity. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105:43-50. http://dx.doi.org/10.1016/j.tripleo.2007.06.011
Hyland PL, Coulter WA, Ruman A, Fulton CR, O`Neill HJ, Coyle PV, et al. Asymptomatic shedding of HSV-1 in patients undergoing oral surgical procedures and attending for noninvasive treatment. Oral Dis. 2007;13:414-8. http://dx.doi.org/10.1111/j.1601-0825.2007.01316.x
Scott DA, Coulter WA, Lamey PJ. Oral shedding of herpes simplex virus tipe 1: A review. J Oral Pathol Med. 1997;26:441-7. http://dx.doi.org/10.1111/j.1600-0714.1997.tb00012.x
Kim HN, Meier A, Huang ML, Kuntz S, Selke S, Celum C, et al. Oral herpes simplex virus type 2 reactivation in HIV-positive and HIV-negative men. J Infect Dis. 2006;194: 420-7. http://dx.doi.org/10.1086/505879
Christensen J, Thomsen A. Coordinating innate and adaptive immunity to viral infection: Mobility is the key. APMIS. 2009;117:338-55. http://dx.doi.org/10.1111/j.1600-0463.2009.02451.x
Bonjardim A. Interferons (IFNs) are key cytokines in both innate and adaptive antiviral immune responses and viruses counteract IFN action. Microbes Infect. 2005;7:569-78. http://dx.doi.org/10.1016/j.micinf.2005.02.001
Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: Mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459-72. http://dx.doi.org/10.1128/ACC.00615-10
Chakrabarti S, Pillay D, Ratcliffe D, Cane PA, Collingham KE, Milligan DW. Resistance to antiviral drugs in herpes simplex virus infections among allogeneic stem cell transplant recipients: Risk factors and prognostic significance. J Infect Dis. 2000;181:2055-8. http://dx.doi.org/10.1086/315524
Danve-Szatanek C, Aymard M, Thouvenot D, Morfin F, Agius G, Bertin I, et al. Surveillance network for herpes simplex virus resistance to antiviral drugs: 3-year follow-up. J Clin Microbiol. 2004;42:242-9. http://dx.doi.org/10.1128/JCM.42.1.242–249.2004.
Bernal LJ, Ávila LV, Marín LM, Casas JA, Bohórquez SP, Barrientos S, et al. Detección de la presencia de antígeno y ADN de virus herpes simplex tipo 1 en ganglios trigeminales humanos. Univ Odontol. 2012;31:117-24.
Manuel O, Panq XL, Humar A, Humar D, Doucette K, Preiksaitis JK. An assessment of donor-to-recipient transmission patterns of human cytomegalovirus by analysis of viral genomic variants. J Infect Dis. 2009;199:1621-8. http://dx.doi.org/10.1086/598952
Chêne A, Nylén S, Donati D, Bejarano MT, Kironde F, Wahlgren M, et al. Effect of acute Plasmodium falciparum malaria on reactivation and shedding of the eight human herpes viruses. PLoS ONE. 2011;6:e26266. http://dx.doi.org/10.1371/journal.pone.0026266
Lucht E, Brytting M, Bjerregaard L, Julander I, Linde A. Shedding of cytomegalovirus and herpesviruses 6, 7, and 8 in saliva of human immunodeficiency virus type 1-infected patients and healthy controls. Clin Infect Dis. 1998;27:137-41. http://dx.doi.org/10.1086/514604
Greenberg MS, Dubin G, Stewart JC, Cumming CG, MacGregor RR, Friedman HM. Relationship of oral disease to the presence of cytomegalovirus DNA in the saliva of AIDS patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79:175-9. http://dx.doi.org/10.1016/S1079-2104(05)80278-1
Sinclair J, Reeves M. The intimate relationship between human cytomegalovirus and the dendritic cell lineage. Front Microbiol. 2014;7:1-14. http://dx.doi.org/10.3389/fmicb.2014.00389
Griffin E, Krantz E, Selke S, Huang ML, Wald A. Oral mucosal reactivation rates of herpes viruses among HIV-1 seropositive persons. J Med Virol. 2008;80:1153-9. http://dx.doi.org/10.1002/jmv.21214
Some similar items:
- Mayra L. Lúa, Arturo Plascencia, Patricia Paredes, Yaxsier de Armas, Miguel Raygoza, Erika Martínez, Fernando Siller, Iván Isidro Hernández, Etiological identification of viral agents in acute encephalitis in Guadalajara, México, 2011-2015 , Biomedica: Vol. 38 No. 2 (2018)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























