Characterization of potential drug-drug interactions in patients hospitalized in the intensive care unit of a tertiary hospital in Bogotá
Abstract
Introduction: Drug-drug interactions occur more frequently in intensive care units than in other services. However, in Colombia, there are few studies on this problem in critically ill patients.
Objectives: To characterize potential drug-drug interactions generated from prescriptions during hospitalization in an intensive care unit and to determine factors associated with their onset.
Materials and methods: A retrospective cohort was assembled with patients hospitalized in an intensive care unit for a seven-month period. The daily prescription was assessed for potential drugdrug interactions using the Lexicomp® program. We calculated the incidence of interactions, classified them by type, severity, and level of documentation, and evaluated the factors associated with their onset using logistic regression.
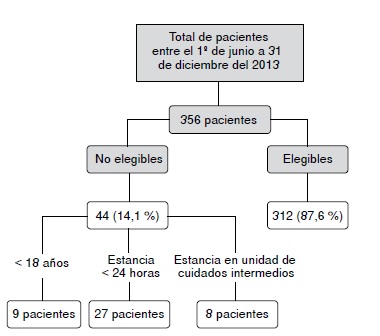
Results: The proportion of patients with at least one interaction was 84% while 87% had more than one interaction; the median was six interactions per patient. The most frequent was fentanyl and midazolam (23%). Moderate interactions were the most frequent by severity (77.6%) and by documentation (52.6%). The most common index and precipitating drugs were midazolam (12%) and fentanyl (10.6%), respectively. Age (OR=3.1) and the number of drugs (OR=11.8) were associated with the occurrence
of interactions.
Conclusions: Given their high frequency and potential negative impact, the systematic monitoring of prescriptions in intensive care units to detect interactions is important. Such monitoring contributes to the rational use of medicines and to improve the quality of care.
Downloads
References
Rodrigues DA, editor. Drug-drug interactions. Second edition. London: Informa Health Care. 2008. p. 768.
Pirmohamed M, James S, Meakin S, Green C, Scout A, Walley T, et al. Adverse drug reactions as cause of admission to hospital: Prospective analysis of 18.820 patients. BMJ. 2004; 329:15-9. https://doi.org/10.1136/bmj.329.7456.15
Pouyanne P, Haramburu F, Imbs J, Bégaud B. Admissions to hospital caused by adverse drug reactions: Cross sectional incidence study. BMJ. 2000;320:1036-45. https://doi.org/10.1136/bmj.320.7241.1036
Becker ML, Kallewaard M, Caspers PW, Visser LE, Leufkens HG, Stricker BH. Hospitalizations and emergency department visits due to drug–drug interactions: A literature review. Pharmacoepidemiol Drug Saf. 2007;16:641-51. https://doi.org/10.1002/pds.1351
Caribé RA, Chaves GR, Pocognoni JD, Souza IA. Potenciales interacciones medicamentosas en pacientes con sepsis internados en la unidad de terapia intensiva. Farm Hosp. 2013;37:383-7. https://doi.org/10.7399/FH.2013.37.5.534
Rivkin A, Yin H. Evaluation of the role of the critical care pharmacist in identifying and avoiding or minimizing significant drug-drug interactions in medical intensive care patients. J Crit Care. 2011;26:104.e1-6. https://doi.org/10.1016/j.jcrc.2010.04.014
Reis AM, Cassiani SH. Prevalence of potential drug interactions in patients in an intensive care unit of a university hospital in Brazil. Clinics (Sao Paulo). 2011;66:9-15. https://doi.org/10.1590/S1807-59322011000100003
Plaza J, Álamo M, Torres P, Fuentes A, López F. Interacciones de medicamentos y eventos adversos en fármacos utilizados en una unidad de cuidados intensivos. Rev Med Chile. 2010;138 452-60. https://doi.org/10.4067/S0034-98872010000400009
Bergk V, Gasse C, Rothenbacher D, Loew M, Brenner H, Haefeli WE. Drug interactions in primary care: Impact of a new algorithm on risk determination. Clin Pharmacol Ther. 2004;76:85-96. https://doi.org/10.1016/j.clpt.2004.02.009
Askari M, Eslami S, Louws M, Wierenga PC, Dongelmans DA, Kuiper RA, et al. Frequency and nature of drug-drug interactions in the intensive care unit. Pharmacoepidemiol Drug Saf. 2013;22:430-7. https://doi.org/10.1002/pds.3415
Smithburger P, Kane-Gill SL, Seybert AL. Drug–drug interactions in the medical intensive care unit: An assessment of frequency, severity and the medications involved. Int J Pharma Pract. 2012;20:402-8. https://doi.org/10.1111/j.2042-7174.2012.00221.x
Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, editors. To err is human: Building a safer health system. Washington (DC): National Academies Press (US); 2000. Fecha de consulta: 17 de febrero de 2017. Disponible en: https://www.ncbi.nlm.nih.gov/books/NBK225182/
Almeida SM, Gama SC, Akamine N. Prevalence and classification of drug-drug interactions in intensive care patients, Einstein (Sao Paulo). 2007;5:347-51.
McFarland HM. Identification and management of drug interactions. Medscape. Fecha de consulta: 20 de mayo de 2016. Disponible en: http://www.medscape.org/viewarticle/418376
Joseph A. Khan A. A comparison of the Micromedex vs. Lexicomp medicine information databases. Fecha de consulta: 20 de mayo de 2016. Disponible en: http://www.
ukmi.nhs.uk/filestore/misc/AcomparisonoftheMicromedexandLexi-compDatabases.pdf
Barrons R. Evaluation of personal digital assistant software for drug interactions. Am J Health Syst Pharm. 2004;61:380-5.
Carvalho RE, Reis AM, Faria LM, ZagoKS, Cassiani SH. Prevalence of drug interactions in intensive care units in Brazil. Acta Paul Enferm. 2013;26:150-7.
Hammes J, Pfuetzenreiter F, daSilveira F, Koenig A, AdrienoWestphal G. Potential drug interactions prevalence in intensive care units. Rev Bras Ter Intensiva. 2008;20:349-54. https://doi.org/10.1590/S0103-507X2008000400006
Cruciol-Souza JM, Thomson JC. Prevalence of potential drug-drug interactions and its associated factors in a Brazilian teaching hospital. J Pharm Pharm Sci. 2006;9:427-33.
Devlin JW, Roberts RJ. Pharmacology of commonly used analgesics and sedatives in the ICU: Benzodiazepines, propofol, and opioids. Crit Care Clin. 2009;25:431-49.
https://doi.org/10.1016/j.ccc.2009.03.003
Riker RR, Fraser GL. Altering intensive care sedation paradigms to improve patient outcomes. Crit Care Clin. 2009;25:527-38. https://doi.org/10.1016/j.ccc.2009.05.004
Schweickert WD, Kress JP. Strategies to optimize analgesia and sedation. Crit Care. 2008;12(Suppl.3):S6. https://doi.org/10.1186/cc6151
López JM. Metabolismo de fármacos: enzimas CYP3A (sub familia del citocromo P450). Fecha de consulta: 21 de marzo de 2013. Disponible en: http://www.info-farmacia.com/bioquimica/metabolismo-de-farmacos-enzimas-cyp3asub-familia-del-citocromo-p450
Hansten PD. Drug interaction management. Pharm World Sci. 2003;25:94-7. https://doi.org/10.1023/A:1024077018902
Elanjian S, Gora ML, Symes LR. Methods used by pharmacy departments to identify drugs interactions. Am J Hosp Pharm. 1993;50:2546-9.
Bucşa C, Farcaş A, Cazacu I, Leucuta D, Achimas-Cadariu A, Mogosan C, et al. How many potential drug–drug interactions cause adverse drug reactions in hospitalized patients? Eur J Intern Med. 2013;24:27-33. https://doi.org/10.1016/j.ejim.2012.09.011
Lima RE, Cassiani SH. Potential drug interactions in intensive care patients at a teaching hospital. Rev Lat Am Enfermagem. 2009;17:222-7. https://doi.org/10.1590/S0104-11692009000200013
El Samia Mohamed SMA, Gad ZM, El-Nimr NA, Abdel Razek AAH. Prevalence and pattern of potential drug-drug interactions in the critical care units of a tertiary hospital in Alexandria, Egypt. Adv Pharmacoepidemiol Drug Saf. 2013;2:144-54. https://doi.org/10.4172/2167-1052.1000144
Some similar items:
- José Roberto Támara-Ramírez, Carlos Arturo Álvarez, Jesús Rodríguez, Loss of follow-up and associated factors in patients enrolled in the HIV/AIDS program of the Hospital Universitario San Ignacio, Colombia, 2012-2013 , Biomedica: Vol. 36 No. 2 (2016)
- Sandra Lorena Girón, Julio César Mateus, Fabián Méndez, Impact of an open waste disposal site on the occurrence of respiratory symptoms and on health care costs of children , Biomedica: Vol. 29 No. 3 (2009)
- Mauricio Beltrán, María Cristina Navas, María Patricia Arbeláez, Jorge Donado, Sergio Jaramillo, Fernando De la Hoz, Cecilia Estrada, Lucía del Pilar Cortés, Amalia de Maldonado, Gloria Rey, Seroprevalence of hepatitis B virus and human immunodeficiency virus infection in a population of multiply-transfused patients in Colombia , Biomedica: Vol. 29 No. 2 (2009)
- Andrés Leonardo González, Ruth Aralí Martínez, Luis Ángel Villar, Clinical evolution of dengue in hospitalized patients , Biomedica: Vol. 28 No. 4 (2008)
- Ligia Inés Moncada, Sandra Milena Rios, Julián Alfredo Fernández, Fabio Rivas, María Luz Sáenz, Pediculosis prevalence and associated risk factors in a nursery school, Bogotá, Colombia , Biomedica: Vol. 28 No. 2 (2008)
- Flor de María Cáceres, Luis Carlos Orozco, Incidence of and factors for non-compliance to antituberculous treatment , Biomedica: Vol. 27 No. 4 (2007)
- Alicia Norma Alayón, Saudith Ariza, Karen Baena, Lina Lambis, Lina Martínez, Lourdes Benítez, Active search and assessment of cardiovascular risk factors in young adults, Cartagena de Indias, 2007 , Biomedica: Vol. 30 No. 2 (2010)
- María del Pilar Chaves, Julián Alfredo Fernández, Isabel Ospina, Myriam Consuelo López, Ligia Moncada, Patricia Reyes, Giardia duodenalis prevalence and associated risk factors in preschool and school-age children of rural Colombia , Biomedica: Vol. 27 No. 3 (2007)
- Elpidia Poveda, Alexandra Cuartas, Saralicia Guarín, Yibby Forero, Elsa Villarreal, Iron and vitamin A micronutrient status, risk factors for their deficiencies and anthropometric assessment in preschool child from Funza municipality, Colombia , Biomedica: Vol. 27 No. 1 (2007)
- Richard Hoyos, Lisandro Pacheco, Luz Adriana Agudelo, German Zafra, Pedro Blanco, Omar Triana, Seroprevalence of Chagas disease and associated risk factors in a population of Morroa, Sucre , Biomedica: Vol. 27 No. 1esp (2007): Enfermedad de Chagas
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























