Quality evaluation of malaria diagnosis in the local laboratories network and in intermediate laboratories in a setting towards the disease elimination in Ecuador
Abstract
Introduction: To reach the goal of malaria elimination in Ecuador for the year 2020, it is necessary to have a laboratory network with the capacity to perform microscopic diagnosis according to the WHO/PAHO quality standards and to provide the adequate treatment of cases.
Objective: To determine the level of competence for parasitological diagnosis of the microscopists from the local public network and the performance of intermediate reference laboratories.
Materials and methods: We conducted a cross-sectional study based on the information collected in workshops carried out to appraise the competence for microscopic diagnosis of the local laboratory network (zonal health coordinating offices 1 to 8) using a slide panel to evaluate diagnosis agreement, as well as the diagnostic performance of the intermediate laboratories using an external quality assessment program. The results were compared against the reference standards of the supranational laboratory in Perú.
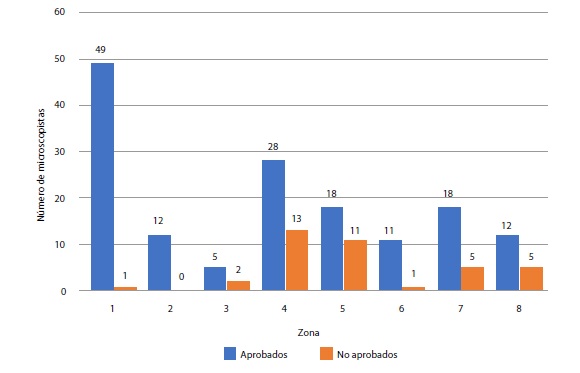
Results: We evaluated the competencies of 191 microscopists in 11 workshops and 153 (80.1%) of them were approved. The medians of the indicators were the following: concordance for parasite detection, 100% (Q1- Q3: 96-100), concordance for species identification, 100% (Q1- Q3: 93-100), and concordances for stage identification, 93.0% (Q1- Q3: 86-95) and parasite counting, 77.0% (Q1- Q3: 71-82). In the external quality assessment, the three intermediate laboratories obtained 100% in parasite detection concordance and 96% for species detection concordance.
Conclusions: The results for the primary network and the performance indicators for the intermediate laboratories showed the high-quality standards of the training program implemented in the country.
Downloads
References
World Health Organization. Eliminating malaria. Geneva: World Health Organization; 2016. Fecha de consulta: 18 de abril de 2018. Disponible en: https://www.who.int/malaria/publications/atoz/eliminating-malaria/en/
Ministerio de Salud Pública de Ecuador. Reformar el reglamento interno para la administración y control de activos fijos del Ministerio de Salud Pública. Fecha de consulta: 20 de abril de 2018. Disponible en: https://aplicaciones.msp.gob.ec/salud/archivosdigitales/documentosDirecciones/dnn/archivos/ac_00005108_2014 02 set.pdf
Presidencia de la República de Ecuador. Decreto Ejecutivo No. 1290/2012 de 30 de agosto. Fecha de consulta: 20 de abril de 2018. Disponible en: http://www.controlsanitario.gob.ec/wpcontent/uploads/downloads/2014/04/DECRETO-EJECUTIVO-DE-CREACION-DEL-ARCSA.pdf
Tangpukdee N, Duangdee C, Wilairatana P, Krudsood S. Malaria diagnosis: A brief review. Korean J Parasitol. 2009;47:93-102. https://doi.org/10.3347/kjp.2009.47.2.93
World Health Organization. Guidelines for the treatment of malaria. Geneva: World Health Organization; 2015. p. 317. Fecha de consulta: 23 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/9789241549127/en/
World Health Organization. Malaria microscopy quality assurance manual. Version 2. Geneva: World Health Organization; 2016. p. 121.
World Health Organization. A framework for malaria elimination. Geneva: World Health Organization; 2017. p. 100. Fecha de consulta: 23 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/9789241511988/en/
Poostchi M, Silamut K, Maude RJ, Jaeger S, Thoma G. Image analysis and machine learning for detecting malaria. Transl Res. 2018;194:36-55. https://doi.org/10.1016/j.trsl.2017.12.004
World Health Organization. Universal access to malaria diagnostic testing: An operational manual. Geneva: WHO; 2011. p. 160. Fecha de consulta: 5 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/9789241502092/en/
World Health Organization. World malaria report, 2017. Geneva: World Health Organization; 2017. p. 196. Fecha de consulta: 5 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/world-malaria-report-2017/en/
Moakofhi K, Edwards JK, Motlaleng M, Namboze J, Butt W, Obopile M, et al. Advances in malaria elimination in Botswana: A dramatic shift to parasitological diagnosis, 2008-2014. Public Health Action. 2018;8:S34-8. https://doi.org/10.5588/pha.17.0017
World Health Organization. Policy brief on malaria diagnostics in low-transmission settings. Fecha de consulta: 25 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/policy-brief-diagnosis-low-transmission-settings/en
Foundation for Innovative New Diagnostics. Malaria rapid diagnostic tests. An implementation guide. Geneva: Foundation for Innovative New Diagnostics; 2013. p. 88. Fecha de consulta: 15 de febrero de 2018. Disponible en: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/243817/malaria_rdt_implementation_guide2013.pdf
Organización Panamericana de la Salud. Informe técnico, cuarto panel, 2014-2015. Programa de evaluación externa del desempeño para el diagnóstico microscópico de la malaria. Fecha de consulta: 27 de abril de 2018. Disponible en: http://www.paho.org/hq/index.php?option=com_docman&task=doc_download&Itemid=&gid=32909&lang=es
Gutiérrez S, Arróspide N. Manual de procedimientos de laboratorio para el diagnóstico de malaria. Lima: Ministerio de Salud, Instituto Nacional de Salud; 2012. p. 46. Fecha de consulta: 15 de abril de 2018. Disponible en: http://www.ins.gob.pe/repositorioaps/0/4/jer/1/Antimalaricos/manualMALARIA.pdf
Organización Panamericana de la Salud. Programa de evaluación externa del desempeño para el diagnóstico microscópico de malaria. Fecha de consulta: 10 de febrero de 2018. Disponible en: http://linksglobal.org/AMI/extras/PEED_Malaria_OPS_Esp_2011.pdf
Carter JY. Special issue: External quality assessment in laboratory medicine. External quality assessment in resource–limited countries. Biochem Med. 2017;27:97-109. https://doi.org/10.11613/BM.2017.013
Yitbarek T, Nega D, Tasew G, Taye B, Desta K. Performance evaluation of malaria microscopists at defense health facilities in Addis Ababa and its surrounding areas, Ethiopia. PLoS One. 2016;11:e0166170. https://doi.org/10.1371/journal.pone.0166170
Subsecretaría Nacional de Vigilancia de la Salud Pública. Enfermedades transmitidas por vectores. Informe malaria. Semana epidemiológica (SE) 01/2/2017. Ecuador. Fecha de consulta: 10 de noviembre de 2018. Disponible en: https://www.salud.gob.ec/wpcontent/uploads/2017/07/Gaceta-Vectorial-SE52.pdf
20. Dirección Nacional de Vigilancia Epidemiológica. Anuario de vigilancia epidemiológica 2017. Enfermedades transmitidas por vectores. Fecha de consulta: 10 de noviembre de 2018. Disponible en: https://public.tableau.com/profile/vvicentee80#!/vizhome/EnfermeddaesTropicales_vectoriales-2014/ANUARIO
Rosas-Aguirre A, Gamboa D, Rodríguez H, Llanos-Zavalaga F, Aguirre K, Llanos-Cuentas A. Uso de paneles de láminas estandarizadas para la evaluación de competencias en el diagnóstico microscópico de malaria en la amazonía peruana. Rev Peru Med Exp Salud Pública. 2010;27:540-7.
Mendoza NM, González NE. Evaluación del desempeño mediante paneles de láminas: una herramienta para la clasificación de los microscopistas senior del Programa de Control de la Malaria en Colombia. Biomédica. 2015;35:582-9. https://doi.org/10.7705/biomedica.v35i4.2694
Sori G, Zewdie O, Tadele G, Samuel A. External quality assessment of malaria microscopy diagnosis in selected health facilities in Western Oromia, Ethiopia. Malar J. 2018;17:233. https://doi.org/10.1186/s12936-018-2386-2
Ashraf S, Kao A, Hugo C, Christophel EM, Fatunmbi B, Luchavez J, et al. Developing standards for malaria microscopy: External competency assessment for malaria microscopists in the Asia-Pacific. Malar J. 2012;11:352. https://doi.org/10.1186/1475-2875-11-352
World Health Organization. Basic malaria microscopy. Part I. Learner´s guide. Geneva: World Health Organization; 2010. p. 83. Fecha de consulta: 10 de abril de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/9241547820/en/26. World Health Organization. Preparation of water buffered to pH 7.2. Malaria microcopy standard operating procedure – MM-SOP-03A. 2016. Fecha de consulta: 17 de enero de 2018. Disponible en: http://www.wpro.who.int/mvp/lab_quality/2096_oms_gmp_sop_03a_rev.pdf
World Health Organization. Preparation of Giemsa working solution. Malaria microscopy standard operating procedure- MM--SOP-04. 2016. Fecha de consulta: 17 de enero de 2018. Disponible en: http://www.wpro.who.int/mvp/lab_quality/2096_oms_gmp_sop_04_rev.pdf
World Health Organization. Cleaning and storing microscope slide. Malaria microscopy standard operating procedure- MM-SOP-01. 2016. Fecha de consulta: 17 de enero de 2018. Disponible en: http://www.wpro.who.int/mvp/lab_quality/2096_oms_gmp_sop_01_rev.pdf
Wanja E, Achilla R, Obare P, Adeny R, Moseti C, Otieno V, et al. Evaluation of a laboratory quality assurance pilot programme for malaria diagnostics in low transmission areas of Kenya, 2013. Malar J. 2017;16:221. https://doi.org/10.1186/s12936-017-1856-2
Biadglegne F, Belyhun Y, Ali J, Walle F, Gudeta N, Kassu A, et al. Does the practice of blood film microscopy for detection and quantification of malaria parasites in northwest Ethiopia fit the standard? BMC Health Serv Res. 2014;14:529. https://doi.org/10.1186/s12913-014-0529-x
Abreha T, Alemayehu B, Tadesse Y, Gebresillassie S, Tadesse A, Demeke L, et al. Malaria diagnostic capacity in health facilities in Ethiopia. Malar J. 2014;13:292. https://doi.org/10.1186/1475-2875-13-292
World Health Organization. Good practices for selecting and procuring rapid diagnostic tests for malaria. Geneva: World Health Organization; 2011. p. 108. Fecha de consulta: 25 de febrero de 2018. Disponible en: http://www.who.int/malaria/publications/atoz/9789241501125/en
Sakandé J, Nikièma A, Kabré E, Sawadogo CH, Nacoulma EW, Sanou M, et al. Implementation of a national external quality assessment program for medical laboratories in Burkina Faso. Am J Clin Pathol. 2014;141:181-7. https://doi.org/10.1309/AJCPXC83HIBBSRDT
Ahmed L, Seal LH, Ainley C, De la Salle B, Brereton M, Hyde K, et al. Web-based virtual microscopy of digitized blood slides for malaria diagnosis: An effective tool for skills assessment in different countries and environments. J Med Internet Res. 2016;18:e213. https://doi.org/10.2196/jmir.6027
Abdul-Ghani R. Towards e-parasitology: Making use of virtual microscopy. Trop Med Int Health. 2015;20:227-9. https://doi.org/10.1111/tmi.12426
Ministerio de Salud Pública. Manual de procedimientos del Subsistema de Vigilancia Epidemiológica Alerta Acción SIVE-ALERTA. Quito: Ministerio de Salud; 2013. Fecha de consulta: 2 de mayo de 2018. Disponible en: https://aplicaciones.msp.gob.ec/salud/archivosdigitales/documentosDirecciones/dnn/archivos/manual_de_procedimientos_sive-alerta.pdf
Sun JL, Zhou S, Geng QB, Zhang Q, Zhang ZK, Zheng CJ, et al. Comparative evaluation of the diagnosis, reporting and investigation of malaria cases in China, 2005–2014: Transition from control to elimination for the national malaria programme. Infect Dis Poverty. 2016;5:65. https://doi.org/10.1186/s40249-016-0163-4
Some similar items:
- Marisol García, Nohora Mendoza, Coordinadores del Programa de Diagnóstico de Malaria, Evaluation of the malaria diagnosis program in the national public laboratory network, Colombia, 1997-1999 , Biomedica: Vol. 22 No. 2 (2002)
- Berlín Londoño, Jaime Carmona, Silvia Blair, Comparison between OptiMAL and the thick smear tests for malaria diagnosis in an endemic area during a non-epidemic period. , Biomedica: Vol. 22 No. 4 (2002)
- Alberto Tobón, Danger signs in the malaria patient , Biomedica: Vol. 29 No. 2 (2009)
- Nohora M. Mendoza, Carlos A. Jaramillo, Felipe Guhl, Julio C. Padilla, Martha C. Rentería, Malaria diagnosis by nested PCR , Biomedica: Vol. 21 No. 4 (2001)
- María Imaz, Sonia Allassia, Mónica Aranibar, Alba Gunia, Susana Poggi, Ana Togneri, Lidia Wolff, Group of Implementation of Fluorescence, Performance of LED fluorescence microscopy for the detection of acid-fast bacilli from respiratory samples in peripheral laboratories in Argentina , Biomedica: Vol. 37 No. 2 (2017)
- Iveth J. González, Metacaspases and their role in the life cycle of human protozoan parasites , Biomedica: Vol. 29 No. 3 (2009)
- Adriana Maldonado-Franco, Luis F. Giraldo-Cadavid, Eduardo Tuta-Quintero, Alirio R. Bastidas, Angélica Moreno-Giraldo , Daniel A. Botero-Rosas, Development of a web application to evaluate spirometry metrics and clinical variables to support COPD diagnosis in primary care , Biomedica: Vol. 44 No. Sp. 1 (2024): Publicación anticipada, Enfermedades crónicas no transmisibles
- Ana María Vásquez, Felipe Sanín, Luis Gonzalo Álvarez, Alberto Tobón, Alexandra Ríos, Silvia Blair, Therapeutic efficacy of a regimen of artesunate-mefloquine-primaquine treatment for Plasmodium falciparum malaria and treatment effects on gametocytic development , Biomedica: Vol. 29 No. 2 (2009)
- Juan Gabriel Piñeros, Margarita Arboleda, Juan Camilo Jaramillo, Silvia Blair, Report of five cases of severe neonatal Plasmodium vivax malaria in Urabá, Colombia , Biomedica: Vol. 28 No. 4 (2008)
- Silvia Blair, Ana Mercedes Rada, Carolina Moreno, Successful in vitro culture of Plasmodium falciparum gametocytes , Biomedica: Vol. 28 No. 4 (2008)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























