un Identification of enteric viruses from raw water using fluoro-immuno-magnetic separation coupled to RT-PCR
Abstract
Introduction: Enteric viruses have been associated with the production of a variety of diseases transmitted by the fecal-oral route and carried through contaminated food and water. Given their structure and composition, they are highly resistant to environmental conditions and most of the chemical agents used in the purification processes. Therefore, the systematic monitoring of raw water is necessary to ensure its quality especially when it is used for producing drinking water for human consumption.
Objective: We identified the presence of rotavirus and hepatitis A virus by means of the fluoro-immuno-magnetic separation technique (FIMS) in raw water taken from four purification plants and their water supplies in the department of Norte de Santander.
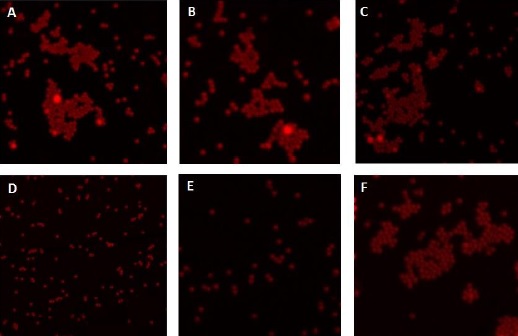
Materials and methods: The viruses were captured and separated from the water samples using magnetic microparticles functionalized with monoclonal anti-Hepatitis A and anti-Rotavirus antibodies. Confocal microscopy was used to monitor the viral concentration process and transmission electron microscopy for the morphological visualization of the separated viruses. The reverse transcriptase-coupled polymerase chain reaction (RT-PCR) was applied to confirm the presence of pathogens.
Results: The two enteric viruses were identified in the majority of the analyzed water samples including water supply sources.
Conclusion: We determined that the FIMS technique together with RT-PCR is highly effective for the detection of viral pathogens in complex matrices such as raw water.
Downloads
References
Organización Mundial de la Salud, Fondo de las Naciones Unidas para la Infancia (UNICEF). Progresos en materia de agua potable, saneamiento e higiene: informe de actualización de 2017 y línea de base de los ods. date of access: march 15, 2019. available from: https://apps.who.int/iris/bitstream/handle/10665/260291/9789243512891-spa.pdf?sequence=1
Ministerio de Agricultura. Decreto 1594 de 1984. “En cuanto a usos del agua y residuos líquidos”. Date of access: April 8, 2019. Available from: www.ideam.gov.co›documents›Dec_1594_1984.pdf
Purparia G, Macalusoa G, Di Bellaa S, Gucciardia F, Miraa F, Di Marco P, et al. Molecular characterization of human enteric viruses in food, water samples, and surface swabs in Sicily. Int J Infect Dis. 2019;80:66-72. https://doi.org/10.1016/j.ijid.2018.12.011
World Health Organization. Guidelines for Drinking-water Quality. Fourth edition. Geneva, 2011. Date of access: April 20, 2019. Available from: whqlibdoc.who.int/publications/2011/9789241548151_eng.pdf
Falman JC, Fagnant-Sperati CS, Kossik AL, Boyle DS, Meschke JS. Evaluation of secondary concentration methods for poliovirus detection in wastewater. Food Environ Virol. 2019;11:20-31. https://doi.org/10.1007/s12560-018-09364-y
Cormier J, Gutiérrez M, Goodridge L, Janes M. Concentration of enteric virus indicator from seawater using granular activated carbon. J Virol Methods. 2014;196:212-8. https://doi.org/10.1016/j.jviromet.2013.11.008
Chandler JC, Schaeffer JW, Davidson M, Magzamen SL, Pérez-Méndez A, Reynolds SJ, et al. A method for the improved detection of aerosolized influenza viruses and the malespecific (F+) RNA coliphage MS2. J Virol Methods. 2017;246:38-41. https://doi.org/10.1016/j.jviromet.2017.04.004
Coughlin BA, Kniel KE. Protocol recovery and detection of enteric viruses from nontraditional irrigation water sources. Methods Protoc. 2019;2:55-63. https://doi.org/10.3390/mps2030055
Haramoto E, Kitajima M, Hata A, Torrey JR, Masago Y, Sano D, et al. A review on recent progress in the detection methods and prevalence of human enteric viruses in water. Water Res. 2018;135:168-86. https://doi.org/10.1016/j.watres.2018.02.004
Ha JH, Choi C, HA SD. Evaluation of immunomagnetic separation method for the recovery of hepatitis A virus and GI.1 and GII.4 Norovirus strains seeded on oyster and mussel. Food Environ Virol. 2014;6:290-6. https://doi.org/10.1007/s12560-014-9156-2
Villamizar R, Osma JF, Ortiz OO. New technique for direct fluoroinmunomagnetic detection of rotavirus in water samples. J Water Health. 2017;15:932-41. https://doi.org/10.2166/wh.2017.028
Peláez D, Guzmán B, Rodríguez J, Acero F, Nava G. Presencia de virus entéricos en muestras de agua para el consumo humano en Colombia: desafíos de los sistemas de abastecimiento. Biomédica. 2016;36:169-78. https://doi.org/10.7705/biomedica.v36i0.2987
Elmahdy EM, Fongaro G, Schissi CD, Petrucio MM, Barardi CRM. Enteric viruses in surface water and sediment samples from the catchment area of Peri Lagoon, Santa Catarina State, Brazil. J Water Health. 2016;14:142-54. https://doi.org/10.2166/wh.2015.295
Environmental Protection Agency. Drinking water contaminant candidate List 4– Draft. EPAHQ-OW-2012-0217. Date of access: August 20, 2017. Available from: http://federalregister.gov/a/2015-02210
García-Alzate C, Valencia CR, Taphorn R, González I. Physicochemical and biological characterization of the Roble River, Upper Cauca, western Colombia. Rev Mus Argent Cienc Nat. 2010;12:5-16. https://doi.org/10.22179/REVMACN.12.247
Rowlands J. Human hepatitis A virus is united with a host of relations. Proc Natl Acad Sci U S A. 2015;112:15010-1. https://doi.org/10.1073/pnas.1520121112
Mathew D, Gautum R. Rotavirus. Clin Lab Med. 2015;35:363-91. https://doi.org/10.1016/j.cll.2015.02.012
Departamento Administrativo Nacional de Estadística (DANE). Censo general 2005. Date of access: June 24, 2019. Available from: https://www.dane.gov.co/index.php/estadisticas-portema/demografia-y-poblacion/censo-general-2005-1
Ministerio de Desarrollo Económico. Dirección de Agua Potable y Saneamiento Básico Reglamento Técnico del Sector de Agua Potable y Saneamiento Básico. RAS 2000. Título B. Date of access: April 8, 2019. Available from: https://www.minvivienda.gov.co/sites/default/files/documentos/titulob-030714.pdf
Forero-Céspedes A, Reinoso-Flórez G. Water quality assessment of the Opia River (Tolima-Colombia), using macroinvertebrates and physicochemical parameters. Caldasia. 2013;35:371-87.
Valverde-Solis A, Moreno-Tamayo E, Ortiz-Palacios NY. Análisis de la calidad de varios cuerpos de aguas superficiales en Bahía Solano utilizando índices de contaminación. Rev Cient Inst Biod Des. 2015;34:14-21. https://doi.org/10.18636/riutch.v34i1.553
Lodder WJ, van den Ber L, Rutjes SA, de Roda Husman AM. Presence of enteric viruses in source waters for drinking water production in the Netherlands. Appl Environ Microbiol. 2010;76:72:17. https://doi.org/10.1128/AEM.00245-10
Koh I, Josephson L. Magnetic nanoparticle sensors. Sensors. 2009;9:8130-45. https://doi.org/10.3390/s91008130
Zhang H, Williams S, Zborowski M, Chalmers J. Binding affinities/avidities of antibody–antigen interactions: Quantification and scale-up implications. Biotechnol Bioeng. 2006;95:5. https://doi.org/10.1002/bit.21024
Center for Diseases Control and Prevention (CDC). Effect of chlorination on inactivating selected pathogen. Date of access: June 28, 2021. Available from: https://www.cdc.gov/safewater/effectiveness-on-pathogens.html
Some similar items:
- María Clara Echeverry, Nubia Catalina Tovar, Guillermo Mora, Presence of antibodies to cardiac neuroreceptors in patients with Chagas disease , Biomedica: Vol. 29 No. 3 (2009)
- Nina Paola Lamprea, Lina María Ortega, Gerardo Santamaría, Ladys Sarmiento, Orlando Torres-Fernández, Production and evaluation of an antiserum for immunohistochemical detection of rabies virus in aldehyde fixed brain tissues , Biomedica: Vol. 30 No. 1 (2010)
- Yenny M. Montenegro-Medina, Luz Aída Rey-Caro, Jurg Niederbacher, Ruth Aralí Martínez-Vega, Fredi Alexander Díaz-Quijano, Luis Ángel Villar-Centeno, Roll of antibodies antiplatelets in viral infection: a systematic review of literature , Biomedica: Vol. 31 No. 1 (2011)
- Juan Carlos Villa-Camacho, Juan Camilo Vargas-Zambrano, John Mario González, Flow cytometry model for the detection of neutralizing antibodies against of IFN-β , Biomedica: Vol. 32 No. 4 (2012)
- Sofía Duque, Rubén Santiago Nicholls, Adriana Arévalo, Rafael Guerrero, Serodiagnosis of giardiasis: identiflcation of immunoglobulin G anti-Giardia duodenalis in sera by ELlSA , Biomedica: Vol. 21 No. 3 (2001)
- Piedad Agudelo Flórez, Marcos Restrepo, María Amparo Lotero, Evaluation of indirect immunofluorescence assay for diagnosis of human leptospirosis. , Biomedica: Vol. 26 No. 2 (2006)
- Beatriz Eugenia Ferro, Ana Lucía Rodríguez, Mauricio Pérez, Bruno Luis Travi, Seroprevalence of Leptospira infection in habitants of peripheral neighborhoods in Cali, Colombia. , Biomedica: Vol. 26 No. 2 (2006)
- Yenny Alviarez, María Lares, Mercedes Viettri, Cruz M. Aguilar, Leidi Herrera, Elizabeth Ferrer, Standardization of a direct agglutination test for the immunodiagnosis of Chagas disease , Biomedica: Vol. 34 No. 2 (2014)
- Mónica Llano, Paula Pavía, Astrid Carolina Flórez, Adriana Cuéllar, John Mario González, Concepción Judith Puerta, Preliminary evaluation of the commercial kit Chagas (Trypanosoma cruzi) IgG-ELISA® in Colombian individuals , Biomedica: Vol. 34 No. 2 (2014)
- Dioselina Peláez, María Cristina Hoyos, Julio César Rendón, Carolina Mantilla, Martha Cecilia Ospina, Fabián Cortés-Mancera, Olga Lucía Pérez, Lady Contreras, Yaneth Estepa, María Patricia Arbeláez, María-Cristina Navas, Hepatitis E virus infection in patients with clinical diagnosis of viral hepatitis in Colombia , Biomedica: Vol. 34 No. 3 (2014)
Funding data
-
Departamento Administrativo de Ciencia, Tecnología e Innovación (COLCIENCIAS)
Grant numbers 112156934429
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























