Cloroquina y sus derivados en el manejo de la COVID-19: una revisión sistemática exploratoria
Resumen
Introducción. Recientemente, investigadores chinos y franceses reportaron la eficacia de la cloroquina y la hidroxicloroquina para inhibir la replicación in vitro del virus SARS-CoV-2. La diseminación oportuna de la información científica es clave en tiempos de pandemia. Es urgente contar con una revisión sistemática sobre el efecto y la seguridad de estos medicamentos en la COVID-19.
Objetivo. Describir el estado actual de la literatura científica publicada hasta el 25 de marzo de 2020 sobre el uso de la cloroquina o sus derivados en el manejo de pacientes con COVID-19.
Materiales y métodos. Se hizo una revisión sistemática exploratoria en PubMed, Embase, Lilacs y 15 bases de datos de la Plataforma de Registros Internacionales de Ensayos Clínicos de la Organización Mundial de la Salud (OMS). Se incluyeron publicaciones empíricas y teóricas en inglés, español, italiano, francés o portugués, y se hizo una síntesis narrativa de los resultados.
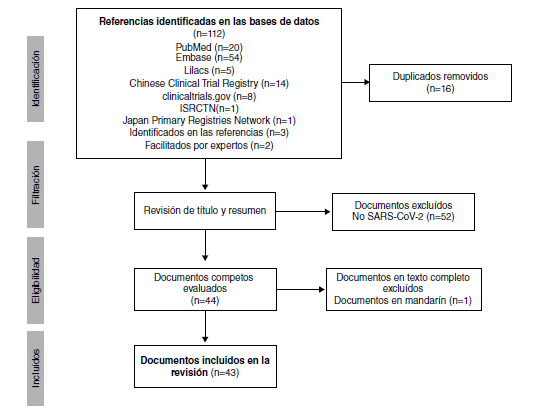
Resultados. Se incluyeron 19 documentos y 24 registros de ensayos clínicos (n=43) de 18.059 pacientes. El 66 % (16/24) de los ensayos están registrados en China. Nueve ensayos evalúan la cloroquina exclusivamente y ocho, la hidroxicloroquina. Los documentos son comentarios (n=9), estudios in vitro (n=3), revisiones narrativas (n=2), guías de práctica clínica (n=2), así como una revisión sistemática, un consenso de expertos y un ensayo controlado.
Conclusiones. Un ensayo clínico pequeño (n=26), no aleatorizado y defectuoso, respalda el uso de la hidroxicloroquina en pacientes con COVID-19. Se requiere de manera urgente tener acceso a los resultados de otros ensayos clínicos para determinar la efectividad y la seguridad de la cloroquina y sus derivados en pacientes con COVID-19.
Descargas
Referencias bibliográficas
World Health Organization. WHO Director-General’s opening remarks at the media briefing on COVID-19. [Fecha de consulta: 23 de marzo de 2020]. Disponible en: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
World Health Organization. Coronavirus disease 2019. [Fecha de consulta: 8 de abril de 2020]. Disponible en: https://www.who.int/emergencies/diseases/novel-coronavirus-2019
Savarino A, Di Trani L, Donatelli I, Cauda R, Cassone A. New insights into the antiviral effects of chloroquine. Lancet Infect Dis. 2006;6:67-9. https://doi.org/10.1016/S1473-3099(06)70361-9
Ksiazek TG, Erdman D, Goldsmith CS, Zaki SR, Peret T, Emery S, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953-66. https://doi.org/10.1056/NEJMoa030781
Keyaerts E, Vijgen L, Maes P, Neyts J, Ranst M Van. In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochem Biophys Res Commun. 2004;323:264-8. https://doi.org/10.1016/j.bbrc.2004.08.085
Yu IT-S, Qiu H, Tse LA, Wong TW. Severe acute respiratory syndrome beyond Amoy gardens: completing the incomplete legacy. Clin Infect Dis. 2014;58:683-6. https://doi.org/10.1093/cid/cit797
Colson P, Rolain J-M, Raoult D. Chloroquine for the 2019 novel coronavirus SARS-CoV-2. Int J Antimicrob Agents. 2020;55:105923. https://doi.org/10.1016/j.ijantimicag.2020.105923
Keyaerts E, Li S, Vijgen L, Rysman E, Verbeeck J, Van Ranst M, et al. Antiviral activity of chloroquine against human coronavirus OC43 infection in newborn mice. Antimicrob Agents Chemother. 2009;53:3416-21. https://doi.org/10.1128/AAC.01509-08
de Wilde AH, Jochmans D, Posthuma CC, Zevenhoven-Dobbe JC, van Nieuwkoop S, Bestebroer TM, et al. Screening of an FDA-approved compound library identifies four small-molecule inhibitors of Middle East respiratory syndrome coronavirus replication in cell culture. Antimicrob Agents Chemother. 2014;58:4875-84. https://doi.org/10.1128/AAC.03011-14
Rolain J-M, Colson P, Raoult D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int J Antimicrob Agents. 2007;30:297-308. https://doi.org/10.1016/j.ijantimicag.2007.05.015
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269-71. https://doi.org/10.1038/s41422-020-0282-0.
Devaux CA, Rolain J-M, Colson P, Raoult D. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int J Antimicrob Agents. 2020;105938. https://doi.org/10.1016/j.ijantimicag.2020.105938
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497-506. https://doi.org/10.1016/S0140-6736(20)30183-5
Shang L, Zhao J, Hu Y, Du R, Cao B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet. 2020;395:683-4. https://doi.org/10.1016/S0140-6736(20)30361-5
Ministerio de Salud y Protección Social - República de Colombia. Hidroxicloroquina y cloroquina se podrán usar para tratamiento de covid – 19. 2020 [Fecha de consulta: 8 de abril de 2020]. Disponible en: https://www.minsalud.gov.co/Paginas/Hidroxicloroquina-y-cloroquina-se-podran-usar-para-tratamiento-de-covid-–-19.aspx
Touret F, de Lamballerie X. Of chloroquine and COVID-19. Antiviral Res. 2020;177:104762. https://doi.org/10.1016/j.antiviral.2020.104762
Song P, Karako T. COVID-19: Real-time dissemination of scientific information to fight a public health emergency of international concern. Biosci Trends. 2020;14:1–2. https://doi.org/10.5582/bst.2020.01056
Arksey H, O’Malley L. Scoping studies: towards a methodological framework. Int J Soc Res Methodol. 2005;8:19-32. https://doi.org/10.1093/geront/gnz021
Levac D, Colquhoun H, O’Brien KK. Scoping studies: advancing the methodology. Implement Sci. 2010;5:69. https://doi.org/10.1186/1748-5908-5-69
World Health Organization. WHO Registry Network. 2020 [Fecha de consulta: 25 de marzo de 2020]. Disponible en: https://www.who.int/ictrp/network/primary/en/
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan—a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. https://doi.org/10.1186/s13643-016-0384-4
Grudniewicz A, Nelson M, Kuluski K, Lui V, Cunningham H V., X Nie J, et al. Treatment goal setting for complex patients: protocol for a scoping review. BMJ Open. 2016;6:e011869. https://doi.org/10.1136/bmjopen-2016-011869.
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann Intern Med. 2018;169:467. https://doi.org/10.7326/M18-0850
Gao J, Tian Z, Yang X. Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020;14:72-73. https://doi.org/10.5582/bst.2020.01047
Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020. https://doi.org/10.1093/cid/ciaa237
Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6:16. https://doi.org/10.1038/s41421-020-0156-0
Gautret P, Lagier J-C, Parola P, Hoang VT, Meddeb L, Mailhe M, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020;105949. https://doi.org/10.1016/j.ijantimicag.2020.105949
Wilson P. COVID-19, Hydroxychloroquine, and the Death of Evidence-Based Medicine. The Methods Man. 2020 [Fecha de consulta: 2 de abril de 2020]. Disponible en: https://www.methodsman.com/blog/covid-19-evidence
Hinton DM. US Food & Drug Administration Emergency Use Autorization for chloroquine phosphate and hydroxychloroquine sulfate use for the treatment of COVID-19. 2020. p. 1–8. [Fecha de consulta: 2 de abril de 2020]. Disponible en: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=f398f8a9-92f3-47cb-81c2-6078806a464d
U.S. Department of Health and Human Services (HHS). HHS accepts donations of medicine to Strategic National Stockpile as possible treatments for COVID-19 patients | HHS.gov. News. 2020 [Fecha de consulta: 2 de abril de 2020]. Disponible en: https://www.hhs.gov/about/news/2020/03/29/hhs-accepts-donations-of-medicine-to-strategic-national-stockpile-as-possible-treatments-for-covid-19-patients.html#
O’Brien N, Hong QN, Law S, Massoud S, Carter A, Kaida A, et al. Health system features that enhance access to comprehensive primary care for women living with hiv in high-income settings: a systematic mixed studies review. AIDS Patient Care STDS. 2018;32:129-48. https://doi.org/10.1089/apc.2017.0305
Alvarado-Castro V, Paredes-Solís S, Nava-Aguilera E, Morales-Pérez A, Alarcón-Morales L, Balderas-Vargas NA, et al. Assessing the effects of interventions for Aedes aegypti control: systematic review and meta-analysis of cluster randomised controlled trials. BMC Public Health. 2017;17(Supl.1):384. https://doi.org/10.1186/s12889-017-4290-z
Shalabi D, Mitchell S, Andersson N. Review of Gender Violence Among Arab Immigrants in Canada: Key Issues for Prevention Efforts. J Fam Violence. 2015;30:817–25. https://doi.org/10.1007/s10896-015-9718-6
Shea BJ, Grimshaw JM, Wells GA, Boers M, Andersson N, Hamel C, et al. Development of AMSTAR: A measurement tool to assess the methodological quality of systematic reviews. BMC Med Res Methodol. 2007;7. https://doi.org/10.1186/1471-2288-7-10
Pimentel J, Arias A, Ramírez D, Molina A, Chomat A-M, Cockcroft A, et al. Game-based learning interventions to foster cross-cultural care training: a scoping review. Games Health J. 2020. https://doi.org/10.1089/g4h.2019.0078.
Pimentel J, Ansari U, Omer K, Gidado Y, Baba MC, Andersson N, et al. Factors associated with short birth interval in low- and middle-income countries: a systematic review. BMC Pregnancy Childbirth. 2020;20:156. https://doi.org/10.1186/s12884-020-2852-z
Peters MDJ, Godfrey CM, Khalil H, McInerney P, Parker D, Soares CB. Guidance for conducting systematic scoping reviews. Int J Evid Based Healthc. 2015;13:141-6. https://doi.org/10.1097/XEB.0000000000000050.
Algunos artículos similares:
- Amanda Maestre, Jaime Carmona-Fonseca, Amanda Maestre, Alta frecuencia de mutaciones puntuales en pfcrt de Plasmodium falciparum y emergencia de nuevos haplotipos mutantes en Colombia , Biomédica: Vol. 28 Núm. 4 (2008)
- Angélica Knudson, Rubén Santiago Nicholls, Ángela Patricia Guerra, Ricardo Sánchez, Perfiles clínicos del paludismo no complicado por Plasmodium falciparum en Córdoba, Colombia , Biomédica: Vol. 27 Núm. 4 (2007)
- Paula Montoya, Alberto Tobón, Silvia Blair, Jaime Carmona, Amanda Maestre, Polimorfismos del gen pfmdr1 en muestras clínicas de Plasmodium falciparum y su relación con la respuesta terapéutica a antipalúdicos y paludismo grave en Colombia , Biomédica: Vol. 27 Núm. 2 (2007)
- Jaime Carmona-Fonseca, Gonzalo Álvarez, Silvia Blair, Malaria por Plasmodium vivax: curación del ataque agudo con tres dosis diferentes de primaquina y dosis fija de cloroquina. Antioquia, Colombia, 2003-2004 , Biomédica: Vol. 26 Núm. 3 (2006)
- Jorge Rivera, Ladys Sarmiento, Edgar Parra, Gabriel Toro, Marcela Neira, Jairo Méndez, Juliana Barbosa, María Leonor Caldas, Alteraciones morfológicas en pulmón por la influenza A H1N1/v09 en autopsias, Colombia, 2009 , Biomédica: Vol. 31 Núm. 3 (2011)
- Manuel Alberto Pérez, Liliana Jazmín Cortés, Ángela Patricia Guerra, Angélica Knudson, Carlos Usta, Rubén Santiago Nicholls, Eficacia de la combinación amodiaquina más sulfadoxinapirimetamina y de la cloroquina para el tratamiento del paludismo en Córdoba, Colombia, 2006 , Biomédica: Vol. 28 Núm. 1 (2008)
- Arletta Añez, Dennis Navarro-Costa, Omar Yucra, Cecilia Garnica, Viviana Melgar, Manuel Moscoso, Ricardo Arteaga, Gladys Nakao, Respuesta terapéutica de Plasmodium vivax a la cloroquina, en Riberalta, Guayaramerín y Yacuiba, Bolivia , Biomédica: Vol. 32 Núm. 4 (2012)
- Lorenzo Cáceres, José Rovira, Rolando Torres, Arsenio García, José Calzada, Manuel De La Cruz, Caracterización de la transmisión de la malaria por Plasmodium vivax en la región fronteriza de Panamá con Costa Rica en el municipio de Barú, Panamá , Biomédica: Vol. 32 Núm. 4 (2012)
- Silvia Blair, Mary Luz López, Juan Gabriel Piñeros, Tania Alvarez, Alberto Tobón, Jaime Carmona, Eficacia terapéutica de tres esquemas de tratamiento de malaria no complicada por Plasmodium falciparum, Antioquia, Colombia, 2002. , Biomédica: Vol. 23 Núm. 3 (2003)
- Eliana Arango, Tania Alvarez, Jaime Carmona, Silvia Blair, Gametocitemia de Plasmodium falciparum según la respuesta terapéutica a sulfadoxina-pirimetamina y cloroquina en dos municipios de Antioquia, Colombia. , Biomédica: Vol. 24 Núm. 1 (2004)
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























