Importancia de la determinación de variantes en el número de copias en neonatos con aneuploidías autosómicas
Resumen
Introducción. Las aneuploidías son trastornos genéticos frecuentes en la práctica clínica; sin embargo, se conoce poco sobre las otras variantes genéticas que modifican el fenotipo final.
Objetivo. Determinar las variantes en el número de copias y las regiones con pérdida de heterocigosidad autosómica mayor de 0,5 % o de regiones mayores de 10 Mb en neonatos con aneuploidías autosómicas.
Materiales y métodos. Se hizo el análisis cromosómico por micromatrices a los neonatos con aneuploidías autosómicas (n=7), trisomía 21 (n=5) y trisomía 18 (n=2) evaluados en los hospitales Antonio Lorena y Regional de Cusco, Perú, en el 2018.
Resultados. En dos neonatos se encontraron variantes en el número de copias, patogénicas o probablemente patogénicas, en regiones diferentes al cromosoma 21 o al 18. Además, se observaron dos variantes del número de copias con más de 500 kpb de patogenia desconocida.
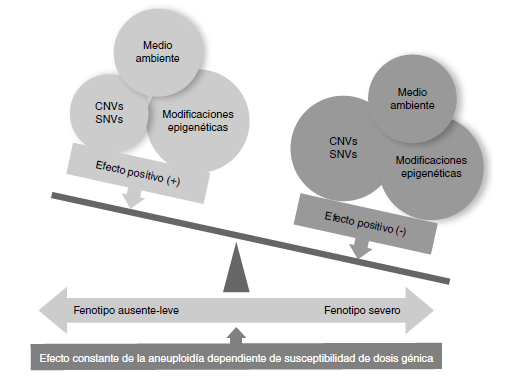
Conclusiones. Si bien el número de pacientes era muy reducido, es importante resaltar que se encontraron otras variantes en el número de copias que se han descrito asociadas con trastornos del neurodesarrollo, varias anomalías congénitas, hipoacusia y talla baja o alta, entre otras, lo que probablemente influye negativamente en el fenotipo de este grupo de pacientes.
Descargas
Referencias bibliográficas
Hutaff-Lee C, Cordeiro L, Tartaglia N. Cognitive and medical features of chromosomal aneuploidy. Handb Clin Neurol. 2013;111:273‑9. https://doi.org/10.1016/B978-0-444-52891-9.00030-0
Nielsen J, Holm V, Haahr J. Prevalence of Edwards’ syndrome. Clustering and seasonal variation? Humangenetik. 1975;26:113‑6. https://doi.org/10.1007/BF00278437
Savva GM, Walker K, Morris JK. The maternal age-specific live birth prevalence of trisomies 13 and 18 compared to trisomy 21 (Down syndrome). Prenat Diagn. 2010;30:57‑64. https://doi.org/10.1002/pd.2403
Sherman SL, Allen EG, Bean LH, Freeman SB. Epidemiology of Down syndrome. Ment Retard Dev Disabil Res Rev. 2007;13:221‑7. https://doi.org/10.1002/mrdd.20157
Abarca-Barriga HH, Chávez-Pastor MA, Trubnykova M, Vásquez F, Poterico JA. Chromosomal microarray analysis in Peruvian children with delayed psychomotor development or intellectual disability. Rev Peru Med Exp Salud Pública. 2017;34:572‑4. https://doi.org/10.17843/rpmesp.2017.343.2741
de Smith AJ, Trewick AL, Blakemore AIF. Implications of copy number variation in people with chromosomal abnormalities: Potential for greater variation in copy number state may contribute to variability of phenotype. Hugo J. 2010;4:1‑9. https://doi.org/10.1007/s11568-010-9144-z
Stranger BE, Forrest MS, Dunning M, Ingle CE, Beazley C, Thorne N, et al. Relative impact of nucleotide and copy number variation on gene expression phenotypes. Science. 2007;315:848‑53. https://doi.org/10.1126/science.1136678
Henrichsen CN, Vinckenbosch N, Zöllner S, Chaignat E, Pradervand S, Schütz F, et al. Segmental copy number variation shapes tissue transcriptomes. Nat Genet. 2009;41:424‑9. https://doi.org/10.1038/ng.345
Girirajan S, Eichler EE. Phenotypic variability and genetic susceptibility to genomic disorders. Hum Mol Genet. 2010;19:R176‑87. https://doi.org/10.1093/hmg/ddq366
Rosenfeld JA, Coe BP, Eichler EE, Cuckle H, Shaffer LG. Estimates of penetrance for recurrent pathogenic copy-number variations. Genet Med. 2013;15:478‑81. https://doi.org/10.1038/gim.2012.164
Schiaffino MC, Bellini C, Costabello L, Caruso U, Jakobs C, Salomons GS, et al. X-linked creatine transporter deficiency: Clinical description of a patient with a novel SLC6A8 gene mutation. Neurogenetics. 2005;6:165‑8. https://doi.org/10.1007/s10048-005-0002-4
Rosenberg C, Freitas ÉL, Uehara DT, Auricchio MTBM, Costa SS, Oiticica J, et al. Genomic copy number alterations in non-syndromic hearing loss. Clin Genet. 2016;89:473‑7. https://doi.org/10.1111/cge.12683
Willatt L, Cox J, Barber J, Cabanas ED, Collins A, Donnai D, et al. 3q29 microdeletion syndrome: Clinical and molecular characterization of a new syndrome. Am J Hum Genet. 2005;77:154‑60. https://doi.org/10.1086/431653
Quintero-Rivera F, Sharifi-Hannauer P, Martinez-Agosto JA. Autistic and psychiatric findings associated with the 3q29 microdeletion syndrome: Case report and review. Am J Med Genet A. 2010;152A:2459‑67. https://doi.org/10.1002/ajmg.a.33573
Carroll LS, Williams HJ, Walters J, Kirov G, O’Donovan MC, Owen MJ. Mutation screening of the 3q29 microdeletion syndrome candidate genes DLG1 and PAK2 in schizophrenia. Am J Med Genet Part B Neuropsychiatr Genet. 2011;156B:844‑9. https://doi.org/10.1002/ajmg.b.31231
Cooper GM, Coe BP, Girirajan S, Rosenfeld JA, Vu T, Baker C, et al. A copy number variation morbidity map of developmental delay. Nat Genet. 2011;43:838‑46. https://doi.org/10.1038/ng.909
Iossifov I, O’Roak BJ, Sanders SJ, Ronemus M, Krumm N, Levy D, et al. The contribution of de novo coding mutations to autism spectrum disorder. Nature. 2014;515:216‑21. https://doi.org/10.1038/nature13908
Warnica W, Merico D, Costain G, Alfred SE, Wei J, Marshall CR, et al. Copy number variable microRNAs in schizophrenia and their neurodevelopmental gene targets. Biol Psychiatry. 2015;77:158‑66. https://doi.org/10.1016/j.biopsych.2014.05.011
Zeng Y, Broxmeyer HE, Staser K, Chitteti BR, Park S-J, Hahn S, et al. Pak2 regulates hematopoietic progenitor cell proliferation, survival, and differentiation. Stem Cells Dayt Ohio. 2015;33:1630‑41. https://doi.org/10.1002/stem.1951
Wang Y, Zeng C, Li J, Zhou Z, Ju X, Xia S, et al. PAK2 haploinsufficiency results in synaptic cytoskeleton impairment and autism-related behavior. Cell Rep. 2018;24:2029‑41. https://doi.org/10.1016/j.celrep.2018.07.061
Singh H, Tiwari P, Bhavi V, Chaudhary PS, Suravajhala P, Mohan MK, et al. Application of chromosomal microarray for evaluation of idiopathic short stature in Asian Indian children: A pilot study. Indian J Endocrinol Metab. 2018;22:100‑6. https://doi.org/10.4103/ijem.IJEM_202_17
Meier N, Bruder E, Lapaire O, Hoesli I, Kang A, Hench J, et al. Exome sequencing of fetal anomaly syndromes: Novel phenotype–genotype discoveries. Eur J Hum Genet. 2019;27:730‑7. https://doi.org/10.1038/s41431-018-0324-y
DECIPHER v9.7: Mapping the clinical genome. Fecha de consulta: 20 mayo de 2016. Disponible en: https://decipher.sanger.ac.uk/browser
Firth HV, Richards SM, Bevan AP, Clayton S, Corpas M, Rajan D, et al. DECIPHER: Database of Chromosomal Imbalance and Phenotype in Humans Using Ensembl Resources. Am J Hum Genet. 2009;84:524‑33. https://doi.org/10.1016/j.ajhg.2009.03.010
Sailani MR, Makrythanasis P, Valsesia A, Santoni FA, Deutsch S, Popadin K, et al. The complex SNP and CNV genetic architecture of the increased risk of congenital heart defects in Down syndrome. Genome Res. 2013;23:1410‑21. https://doi.org/10.1101/gr.147991.112
Rambo-Martin BL, Mulle JG, Cutler DJ, Bean LJH, Rosser TC, Dooley KJ, et al. Analysis of copy number variants on chromosome 21 in Down syndrome-associated congenital heart defects. G3 (Bethesda). 2017;8:105‑11. https://doi.org/10.1534/g3.117.300366
Mégarbané A, Noguier F, Stora S, Manchon L, Mircher C, Bruno R, et al. The intellectual disability of trisomy 21: Differences in gene expression in a case series of patients with lower and higher IQ. Eur J Hum Genet. 2013;21:1253‑9. https://doi.org/10.1038/ejhg.2013.24
Park J, Chung KC. New perspectives of Dyrk1A role in neurogenesis and neuropathologic features of Down syndrome. Exp Neurobiol. 2013;22:244‑8. https://doi.org/10.5607/en.2013.22.4.244
Rodríguez-Cadilla MR. Consideraciones bioéticas y jurídicas de la información genética y el diagnóstico prenatal. Vox Juris. 2015;28:15‑40.
Abarca-Barriga H. Perfil epidemiológico de las anomalías genéticas y congénitas en el Servicio de Citogenética y Citopatología del Hospital Nacional Guillermo Almenara Irigoyen (tesis). Lima: Universidad Nacional Mayor de San Marcos; 2007.
Carreira IM, Ferreira SI, Matoso E, Pires LM, Ferrão J, Jardim A, et al. Copy number variants prioritization after array-CGH analysis – a cohort of 1000 patients. Mol Cytogenet. 2015;8:103‑11. https://doi.org/10.1186/s13039-015-0202-z
Riggs ER, Andersen EF, Cherry AM, Kantarci S, Kearney H, Patel A, et al. Technical standards for the interpretation and reporting of constitutional copy-number variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics (ACMG) and the Clinical Genome Resource (ClinGen). Genet Med. 2019;22:245-57. https://doi.org/10.1038/s41436-019-0686-8
Fan Y-S, Ouyang X, Peng J, Sacharow S, Tekin M, Barbouth D, et al. Frequent detection of parental consanguinity in children with developmental disorders by a combined CGH and SNP microarray. Mol Cytogenet. 2013;6:38‑43. https://doi.org/10.1186/1755-8166-6-38
Rinaldi B, van Hoof E, Corveleyn A, van Cauter A, de Ravel T. BCAP31-related syndrome: The first de novo report. Eur J Med Genet. 2019;103732. https://doi.org/10.1016/j.ejmg.2019.103732
Sayee R, Thomas IM. Consanguinity, non-disjunction, parental age and Down’s syndrome. J Indian Med Assoc. 1998;96:335‑7.
Algunos artículos similares:
- Juan Gabriel Piñeros, Margarita Arboleda, Juan Camilo Jaramillo, Silvia Blair, Reporte de cinco casos de malaria neonatal grave por Plasmodium vivax en Urabá, Colombia , Biomédica: Vol. 28 Núm. 4 (2008)
- Yolanda Cifuentes, Isabel De la Hoz, Martha Bermúdez, Clara Arteaga, Acidemia orgánica (propiónica) en un neonato detectada por espectrometría de masas en tándem , Biomédica: Vol. 28 Núm. 1 (2008)
- Juan Carlos Herrera, Luis Fernando Isaza, José Luis Ramírez, Gonzalo Vásquez, Carlos Mario Muñetón, Detección de aneuploidías del cromosoma 17 y deleción del gen TP53 en una amplia variedad de tumores sólidos mediante hibridación in situ fluorescente bicolor , Biomédica: Vol. 30 Núm. 3 (2010)
- Daniel Echeverri, Dhayra Karem Barreto, Lyda Osorio, Armando Cortés, Ernesto Martínez, Malaria por Plasmodium vivax transmitida por transfusión de un donante asintomático a un recién nacido prematuro , Biomédica: Vol. 32 (2012): Suplemento 1, Malaria
- Husein Husein-El Ahmed, Guillermo Arturo Cañadas-De la Fuente, Rafael Fernández-Castillo, Emilio González-Jiménez, Jesús Cantero-Hinojosa, Marita Lardón-Fernández, Candidiasis cutánea generalizada en recién nacido a término , Biomédica: Vol. 32 Núm. 2 (2012)
- Doris Martha Salgado, Jairo Antonio Rodríguez, Liliana del Pilar Lozano, Tatiana Esther Zabaleta, Dengue perinatal , Biomédica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Yolanda Cifuentes, Martha Isabel Murcia, Jorge Piar, Patricia Pardo, Microcalcificaciones cerebrales en un recién nacido con tuberculosis congénita , Biomédica: Vol. 36 Núm. 1 (2016)
- Claudia Marcela Muñoz, José Orlando Castillo, Daniela Salas, Milena Alexandra Valderrama, Claudia Teresa Rangel, Heiddy Patricia Vargas, Diana Carolina Silva, Manifestaciones mucocutáneas atípicas por fiebre por el virus del chikungunya en neonatos y lactantes de Cúcuta, Los Patios y Villa del Rosario, Norte de Santander, Colombia, 2014 , Biomédica: Vol. 36 Núm. 3 (2016)
- Eliana Patricia Calvo, Carolina Coronel-Ruiz, Syrley Velazco, Myriam Velandia-Romero, Jaime E. Castellanos, Diagnóstico diferencial de dengue y chikungunya en pacientes pediátricos , Biomédica: Vol. 36 (2016): Suplemento 2, Enfermedades virales
- Juan Sebastián Peinado-Acevedo, Estephanía Chacón-Valenzuela, Laura Liliana Rodríguez-Moncada, Síndrome del bebé bronceado, una complicación impredecible de la fototerapia , Biomédica: Vol. 38 Núm. Sup.1 (2018): Suplemento 1, Enfermedades crónicas
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























