Sarcopenia y albúmina sanguínea: revisión sistemática con metaanálisis
Resumen
La sarcopenia se caracteriza por la pérdida de musculatura durante el envejecimiento, lo que puede traer consecuencias para la salud. Se detecta de diversas formas, una de ellas, el uso de biomarcadores sanguíneos como la albúmina, aunque todavía no se ha establecido dicha asociación de forma definitiva.
Esta revisión sistemática y metaanálisis resume el conocimiento sobre la asociación entre sarcopenia y albúmina sérica en los adultos mayores, centrada en la etiología y los factores de riesgo. La revisión se hizo utilizando el programa del Joanna Briggs Institute y la búsqueda incluyó las bases de datos Medline, Embase, CINAHL, y LILACS; la búsqueda manual estuvo a cargo de dos revisores de forma independiente. Para el metaanálisis, se utilizó el programa EpiDat, versión 3.1; las diferencias de medias en los puntajes de lbúmina desagregados por sarcopenia se analizaron mediante el modelo de efectos aleatorios. El grado de heterogeneidad se evaluó con la prueba Q de DerSimonian y Laird.
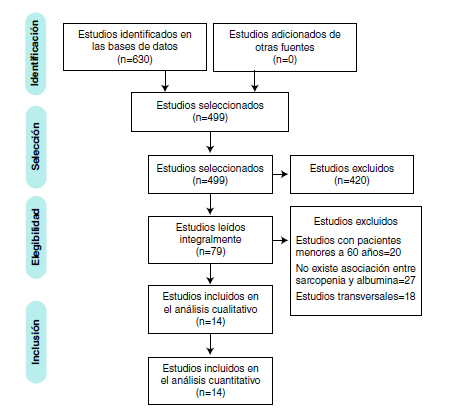
Se analizaron 630 artículos, de los cuales 14 se incluyeron en la revisión. En el metaanálisis, se evidenciaron mayores cantidades de albúmina sanguínea en los adultos mayores que no presentaron sarcopenia frente a los que sí, una diferencia con significación estadística. Aunque hay estudios en los que se explora la asociación entre albúmina y sarcopenia, persiste la necesidad de evaluar la asociación entre los marcadores biológicos, comparándolos entre sí para determinar cuáles pueden utilizarse en la detección de sarcopenia en el adulto mayor.
Descargas
Referencias bibliográficas
United Nations. World population aging 2019: Highlights. 2019. Fecha de consulta: 14 de marzo de 2021. Disponible en: https://www.un.org/en/development/desa/population/publications/pdf/ageing/WorldPopulationAgeing2019-Highlights.pdf
Organización Mundial de la Salud. Informe Mundial sobre el envejecimiento y la salud. 2015. Fecha de consulta: 16 de marzo de 2021. Disponible en: https://apps.who.int/iris/bitstream/handle/10665/186466/9789240694873_spa.pdf?sequence=1
Larsson L, Degens H, Li M, Salviati L, Lee YI, Thompson W, et al. Sarcopenia: Aging-related loss of muscle mass and function. Physiol Rev. 2019;99:427-511. https://doi.org/10.1152/physrev.00061.2017
Wilkinson M, Piasecki O, Atherton PJ. The age-related loss of skeletal muscle mass and function: Measurement and physiology of muscle fiber atrophy and muscle fiber loss in humans. J Ageing Res Rev. 2018;47:123-32. https://doi.org/10.1016/j.arr.2018.07.005
Cruz-Jentoft AJ, Bahat G, Bauer J, Bore Y, Bruyère O, Cederholm T, et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16-31. https://doi.org/10.1093/ageing/afy169
Barbosa-Silva TG, Bielemann RM, González MC, Menezes AMB. Prevalence of sarcopenia among community-dwelling elderly of a medium-sized South American city: Results of the COM VAI? Study study. J Cachexia Sarcopenia Muscle. 2016;7:136-43. https://doi.org/10.1002/jcsm.12049
Cabrerizo S, Cuadras D, Gomez-Busto F, Artaza-Artabe I, Marín-Ciancas F, Malafarina V. Serum albumin and health in older people: Review and meta-analysis. Maturitas. 2015;81:17-27. https://doi.org/10.1016/j.maturitas.2015.02.009
Keller U. Nutritional laboratory markers in malnutrition. J Clin Med. 2019;8:775. https://doi.org/10.3390/jcm8060775
Zhang Z, Pereira SL, Luo M, Matheson EM. Evaluation of blood biomarkers associated with risk of malnutrition in older adults: A systematic review and meta-analysis. Nutrients. 2017;9:829. https://doi.org/10.3390/nu9080829
Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, et al. Systematic reviews of etiology and risk. In: Aromataris E, Munn Z, editors. JBI Manual for Evidence Synthesis. JBI, 2020. Fecha de consulta: 14 de abril de 2021. Disponible en: https://synthesismanual.jbi.global
Aromataris E, Munn Z. JBI Systematic Reviews. In: Aromataris E, Munn Z, editors. Joanna Briggs Institute Reviewer’s Manual. The Joanna Briggs Institute. 2017. Fecha de consulta: 8 de noviembre de 2020. Disponible en: https://synthesismanual.jbi.globalhttps://wiki.joannabriggs.org/display/MANUAL/
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analysis: The PRISMA statement. Ann Inter Med. 2009;151:264-9. https://doi.org/10.1371/journal.pmed.1000097
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for nonrandomized studies (minors): Development and validation of a new instrument. ANZ J Surg. 2003;73:712-6. https://doi.org/10.1046/j.1445-2197.2003.02748.x
Abbas H, Perna S, Shah A, Al-Mannai M, Gasparri VI, Cereda E, et al. Risk factors for 5-year mortality in a cohort of elderly patients with sarcopenia. Exp Gerontol. 2020;136:110944. https://doi.org/10.1016/j.exger.2020.110944
Gong G, Wan W, Zhang X, Liu Y, Liu X, Yin J. Correlation between the Charlson comorbidity index and skeletal muscle mass/physical performance in hospitalized older people potentially suffering from sarcopenia. BMC Geriatr. 2019;19:367. https://doi.org/10.1186/s12877-019-1395-5
D’Alessandro C, Piccoli GB, Barsotti M, Tassi S, Giannese D, Morganti R, et al. Prevalence and correlates of sarcopenia among elderly CKD outpatients on tertiary care. Nutrients. 2018;10:1951. https://doi.org/10.3390/nu10121951
Gariballa S, Alessa A. Association between nutritional blood-based biomarkers and clinical outcome in sarcopenia patients. Clin Nutr ESPEN. 2018;25:145-8. https://doi.org/10.1016/j.clnesp.2018.03.002
Yoo J, Ha Y, Choi H, Kim K, Lee Y, Koo K, et al. Malnutrition and chronic inflammation as risk factors for sarcopenia in elderly patients with hip fracture. Asia Pac J Clin Nutr. 2018;27:527-32. https://doi.org/10.6133/apjcn.082017.02
Zhou C, Zhang F, Zhang F, Yu Z, Chen X, Shen X, et al. Sarcopenia: A new predictor of postoperative complications for elderly gastric cancer patients who underwent radical gastrectomy. J Surg Res. 2017;211:137-46. https://doi.org/10.1016/j.jss.2016.12.014
Harimoto N, Yoshizumi T, Shimokawa M, Sakata K, Kimura K, Itoh SI, et al. Sarcopenia is a poor prognostic factor following hepatic resection in patients aged 70 years and older with hepatocellular carcinoma. Hepatol Res. 2016;46:1247-55. https://doi.org/10.1111/hepr.12674
Hirasawa Y, Nakashima J, Yunaima D, Sugihara T, Gondo T, Nakagami Y, et al. Sarcopenia as a novel preoperative prognostic predictor for survival in patients with bladder cancer undergoing radical cystectomy. Ann Surg Oncol. 2016;23(Suppl.5):1048-54. https://doi.org/10.1245/s10434-016-5606-4
Ishihara H, Kondo T, Omac K, Takagi T, Iizuka J, Kobayashi H, et al. Sarcopenia and the modified Glasgow prognostic score are significant predictors of survival among patients with metastatic renal cell carcinoma who are receiving first-line sunitinib treatment. Target Oncol. 2016;11:605-17. https://doi.org/10.1007/s11523-016-0430-0
Sugimoto T, Ono R, Murta S, Saji N, Matsui Y, Nuda S, et al. Prevalence and associated factors of sarcopenia in elderly subjects with amnestic mild cognitive impairment on Alzheimer disease. Curr Alzheimer Res. 2016;13:718-26. https://doi.org/10.2174/1567205013666160211124828
Wang S, Zhuang C, Huang D, Pang W, Lou N, Chen F, et al. Sarcopenia adversely impacts postoperative clinical outcomes following gastrectomy in patients with gastric cancer: A prospective study. Ann Surg Oncol. 2016;23:556-64. https://doi.org/10.1245/s10434-015-4887-3
Kim H, Suzuki T, Kim M, Kojima N, Yoshida Y, Hirano H, et al. Incidence and predictors of sarcopenia onset in community-dwelling elderly Japanese women: 4-year follow-up study. J Am Med Dir Assoc. 2015;16:85.e1-8. https://doi.org/10.1016/j.jamda.2014.10.006
Gariballa S, Alessa A. Sarcopenia: Prevalence and prognostic significance in hospitalized patients. Clin Nutr. 2013;32:772-6. https://doi.org/10.1016/j.clnu.2013.01.010
Harimoto N, Shirabe K, Yamashita YI, Ikemagi T, Yoshizumi T, Soejima Y, et al. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg. 2013;100:1523-30. https://doi.org/10.1002/bjs.9258
Picca A, Calvani R. Molecular mechanism and pathogenesis of sarcopenia: An overview. Int J Mol Sci. 2021;22:3032. https://doi.org/10.3390/ijms22063032
Cruz-Jentoft AJ, Sayer AA. Sarcopenia. Lancet. 2019;393:2636-46. https://doi.org/10.1016/S0140-6736(19)31138-9
Atherton PJ, Etheridge T, Watt PW, Wilkinson D, Selby A, Rankin D, et al. Muscle full effect after oral protein: Time-dependent concordance and discordance between human muscle protein synthesis and mTORC1 signaling. Am J Clin Nutr. 2010;92:1080-8. https://doi.org/10.3945/ajcn.2010.29819
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16-31. https://doi.org/10.1093/ageing/afy169
Maden-Wilkinson TM, Degens H, Jones DA, McPhee JS. Comparison of MRI and DXA to measure muscle size and age-related atrophy in thigh muscles. J Musculoskelet Neuronal Interact. 2013;13:320-8.
Lee RD, Nieman DC. Nutritional Assessments. North Carolina: Science Engineering, McGraw-Hill; 2002. p.1648.
Shafiee G, Keshtkar A, Soltani A, Ahadi Z, Larijani B, Heshmat R. Prevalence of sarcopenia in the world: A systematic review and meta-analysis of general population studies. Diabetes Metab Disord. 2017;16:21. https://doi.org/10.1186/s40200-017-0302-x
Shin MJ, Jeon YK, Kin IJ. Testosterone and sarcopenia. World J Mens Health. 2018;36:192-8. https://doi.org/10.5534/wjmh.180001
Bian A, Zhou X, Guo Y, Wang W, Zhang Y, Wang X. Association between sarcopenia and levels of growth hormone and insulin-like growth factor-1 in the elderly. BMC Musculoskelet Disord. 2020;21:214. https://doi.org/10.1186/s12891-020-03236-y
Sánches-Castellano C, Matín-Aragón S, Bermejo-Bescós P, Vaquero-Pinto N, Miret-Conchado C, Miguel AM, et al. Biomarkers of sarcopenia in very old patients with hip fracture. J Cachexia Sarcopenia Muscle. 2020;11:478-86. https://doi.org/10.1002/jcsm.12508
Walston J, Mc Burnie MA, Newman A, Tracy RP, Kop CH. Hirsch J, et al. Frailty and activation of the inflammation and coagulation systems with and without clinical comorbidities: Results from the cardiovascular health study. Arch Inter Med. 2002;162:2333-41. https://doi.org/10.1001/archinte.162.20.2333
Ross AC, Caballero B, Cousins RJ, Tucker KL, Zigler TR. Modern Nutrition. In: Health and disease. 11° edition. Philadelphia: Wolters Kluwer; 2014. p. 16-24.
van Atteveld V, van Ancum JM, Reijnierse EM, Trappenburg MC, Meskers CGM, Maier AB. Erythrocyte sedimentation rate and albumin as markers of inflammation are associated with measures of sarcopenia: A cross-sectional study. BMC Geriatr. 2019;19:233. https://doi.org/10.1186/s12877-019-1253-5
Smith S. Using albumin and prealbumin to assess nutritional status. Nursing. 2017;47:65-6. https://doi.org/10.1097/01.NURSE.0000511805.83334.df
Valenzuela RE, Ponce JA, Morales-Figueroa GG, Muro KA, Carreon VR, Aleman-Mateo H. Insufficient amounts and inadequate distribution of dietary protein intake in apparently healthy older adults in a developing country: Implications for dietary strategies to prevent sarcopenia. Clin Interv Aging. 2013;8:1143-8. https://doi.org/10.2147/CIA.S49810
Kragh-Hansen U. Human serum albumin: A multifunctional protein. In: Otagiri M, Chuang V, editor. Albumin in medicine. Singapore: Springer; 2016. p. 1-24. https://doi.org/10.1007/978-981-10-2116-9_1
Algunos artículos similares:
- Karen Luna-Orozco, Julián Alfredo Fernández-Niño, Claudia Iveth Astudillo-García, Asociación entre la discapacidad física y la incidencia de síntomas depresivos en adultos mayores mexicanos , Biomédica: Vol. 40 Núm. 4 (2020)
- Carmen Lucía Curcio, Andrés Fernando Giraldo, Fernando Gómez, Fenotipo de envejecimiento saludable de personas mayores en Manizales , Biomédica: Vol. 40 Núm. 1 (2020)
- Johan Sebastián Villada-Gómez, Clara Helena González-Correa, Felipe Marulanda-Mejía, Puntos de corte provisionales para el diagnóstico de sarcopenia en ancianos de Caldas, Colombia , Biomédica: Vol. 38 Núm. 4 (2018)
- Doris Cardona, Ángela Segura, Alejandra Segura, Diana Muñoz, Daniel Jaramillo, Douglas Lizcano, Maite Catalina Agudelo, Catalina Arango, Santiago Morales, Índice de vulnerabilidad de adultos mayores en Medellín, Barranquilla y Pasto , Biomédica: Vol. 38 Núm. Sup.1 (2018): Suplemento 1, Enfermedades crónicas
- Paola Andrea Filigrana, Olga Lucía Gómez, Fabián Méndez, Impacto de un sitio de disposición final de residuos sólidos en la salud respiratoria de los adultos mayores , Biomédica: Vol. 31 Núm. 3 (2011)
- Alejandro Estrada, Doris Cardona, Ángela María Segura, Lina Marcela Chavarriaga, Jaime Ordóñez, Jorge Julián Osorio, Calidad de vida de los adultos mayores de Medellín , Biomédica: Vol. 31 Núm. 4 (2011)
- Edison Pineda , Alejandra Fernández, Carmen Lucía Curcio, Juliana Fernandes de Souza, Afshin Vafaei, José Fernando Gómez, Medicación potencialmente inapropiada en adultos mayores de la comunidad: análisis longitudinal del estudio IMIAS , Biomédica: Vol. 44 Núm. 2 (2024)
- Marlen Cháves, Caracterización de reacciones adversas a medicamentos en adultos mayores de 44 años en Bogotá, D.C., enero a diciembre, 2012 , Biomédica: Vol. 35 Núm. 1 (2015)
- Doris Cardona, Alejandra Segura, Ángela Segura, María Osley Garzón, Efectos contextuales asociados a la variabilidad del riesgo de depresión en adultos mayores, Antioquia, Colombia, 2012 , Biomédica: Vol. 35 Núm. 1 (2015)
- Ángela Quintero, María Eucaris Henao, María Mercedes Villamil, Jairo León, Cambios en la depresión y el sentimiento de soledad después de la terapia de la risa en adultos mayores internados , Biomédica: Vol. 35 Núm. 1 (2015)
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |

























