A case series of pure neural leprosy in patients diagnosed in a specialized center for the control of Hansen’s disease in Colombia
Abstract
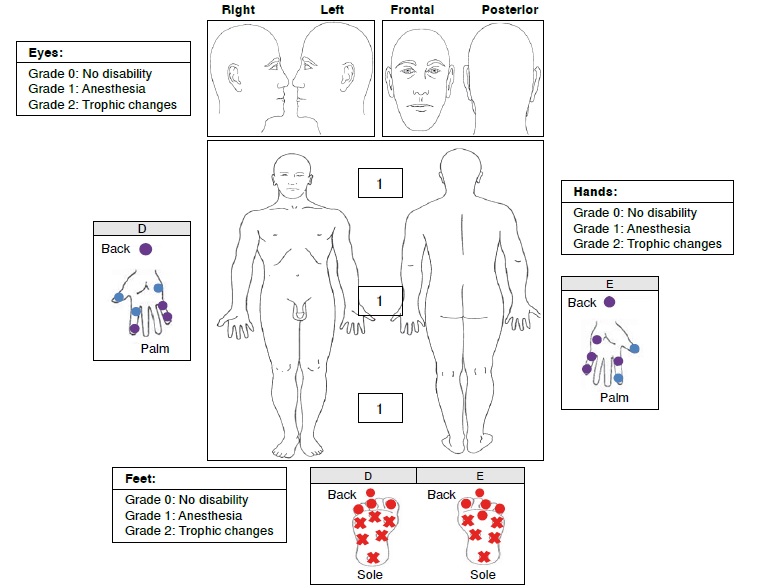
Pure neural leprosy, defined as a peripheral neuropathy in which the patient has no skin lesions, is difficult to diagnose. Its verification by bacteriological index and histopathology is not possible in the majority of the patients.
We describe four cases of pure neural leprosy diagnosed by clinical criteria. The clinical outcome of three of the patients after specific treatment was satisfactory, while the other one developed progressive neural damage despite the therapy. All patients were treated in a specialized center for the management and control of Hansen’s disease in the municipality of Contratación, Santander, Colombia.
Downloads
References
Rodríguez G, Pinto R. Lepra neural primaria: definición y criterios de manejo. Rev Asoc Colomb Dermatol. 2010;18:91-5.
Payne R, Baccon J, Dossett J, Scollard D, Byler D, Patel A, et al. Pure neuritic leprosy presenting as ulnar nerve neuropathy: A case report of electrodiagnostic, radiographic, and histopathological findings. J Neurosurg. 2015;123:1238-43. https://doi.org/10.3171/2014.9.JNS142210
Garbino JA, Marques W, Barreto JA, Heise CO, Rodrigues MM, Antunes SL, et al. Primary neural leprosy: Systematic review. Arq Neuropsiquiatr. 2013;71:397-404.
https://doi.org/10.1590/0004-282X20130046
Reja AH, De A, Biswas S, Chattopadhyay A, Chatterjee G, Bhattacharya B, et al. Use of fine needle aspirate from peripheral nerves of pure-neural leprosy for cytology and PCR to confirm the diagnosis: A pilot study. Indian J Dermatol Venereol Leprol. 2013;79:789-94. https://doi.org/10.4103/0378-6323.120731
Sharma R, Lahiri R, Scollard DM, Pena M, Williams DL, Adams LB, et al. The armadillo: A model for the neuropathy of leprosy and potentially other neurodegenerative diseases. Dis Model Mech. 2013;6:19-24. https://doi.org/10.1242/dmm.010215
Nascimento OJ. Leprosy neuropathy: Clinical presentations. Arq Neuropsiquiatr. 2013;71:661-6. https://doi.org/10.1590/0004-282X20130146
Chacha JJ, Sotto MN, Peters L, Lourenço S, Rivitti EA, Melnikov P. Peripheral nervous system and grounds for the neural insult in leprosy. An Bras Dermatol. 2009;84:495-500. https://doi.org/10.1590/S0365-05962009000500008
Rodríguez G, Pinto R, Gómez Y, Rengifo ML, Estrada OL, Sarmiento M, et al. Pure neuritic leprosy in patients from a high endemic region of Colombia. Lepr Rev. 2013;84:41-50.
Jardim MR, Antunes SL, Santos AR, Nascimento OJ, Nery JA, Sales AM, et al. Criteria for diagnosis of pure neural leprosy. J Neurol. 2003;250:806-9. https://doi.org/10.1007/s00415-003-1081-5
Jardim MR, Antunes SL, Simons B, Wildenbeest JG, Nery JA, Illarramendi X, et al. Role of PGL-I antibody detection in the diagnosis of pure neural leprosy. Lepr Rev. 2005;76:232-40.
Sehgal VN, Sardana KS. «Intriguing» repercussions of primary neuritic leprosy during the evolution of leprosy across the leprosy spectrum. Int J Dermatol. 2006;45:1121-3. https://doi.org/10.1111/j.1365-4632.2006.02882.x
Hui M, Uppin MS, Challa S, Meena AK, Kaul S. Pure neuritic leprosy: Resolving diagnostic issues in acid fast bacilli (AFB)-negative nerve biopsies: A single centre experience from South India. Ann Indian Acad Neurol. 2015;18:292-7. https://doi.org/10.4103/0972-2327.162284
Antunes SL, Chimelli L, Jardim MR, Vital RT, Nery JA, Corte-Real S, et al. Histopathological examination of nerve samples from pure neural leprosy patients: Obtaining maximum information to improve diagnostic efficiency. Mem Inst Oswaldo Cruz. 2012;107:246-53. https://doi.org/10.1590/S0074-02762012000200015
Theuvenet WJ, Miyazaki N, Roche P, Shrestha I. Cytological needle aspiration of the nerve for the diagnosis of pure neural leprosy. Int J Lepr Other Mycobact Dis. 1993;61:597-9.
Grossi MA, Leboeuf MA, Andrade AR, Bührer-Sékula S, Antunes CM. Risk factors for ML Flow seropositivity in leprosy patients. Rev Soc Bras Med Trop. 2008;41(Suppl.2):39-44. https://doi.org/10.1590/S0037-86822008000700009
Ministerio de Salud y Protección Social. Guía de Atención Integral de la Lepra. 2012. Fecha de consulta: 23 de abril de 2016. Disponible en: http://www.minsalud.gov.co/Documentos%20y%20Publicaciones/GUIA%20DE%20ATENCI%C3%93N%20DE%20LEPRA.pdf
Cardona-Castro N, Beltrán JC, Ortiz A, Vissa V. Detection of Mycobacterium leprae DNA in nine-banded armadillos (Dasypus novemcinctus) from the Andean region of Colombia. Lepr Rev. 2009;80:424-31.
Truman RW, Singh P, Sharma R, Busso P, Rougemont J, Paniz-Mondolfi A, et al. Probable zoonotic leprosy in the southern United States. N Engl J Med. 2011;364:1626-33. https://doi.org/10.1056/NEJMoa1010536
Talhari S, Garrido N, Oliveira G, Leide M. Hanseníase. 4.a ed. Manaus: Gráfica Tropical; 2006. p. 216.
Clark BM, Murray CK, Horvath LL, Deye GA, Rasnake MS, Longfield RN. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78:962-7.
Smith WC, Anderson AM, Withington SG, van Brakel WH, Croft RP, Nicholls PG, et al. Steroid prophylaxis for prevention of nerve function impairment in leprosy: Randomised placebo controlled trial (TRIPOD 1). BMJ. 2004;328:1459. https://doi.org/10.1136/bmj.38107.645926.AE
Some similar items:
- Gerzaín Rodríguez, Rafael Henriquez, Shirley Gallo, César Panqueva, Histoid leprosy with giant lesions of fingers and toes , Biomedica: Vol. 35 No. 2 (2015)
- Ismael de Jesús Yepes, Beatriz Lince, Clara Caez, Giovanni de Vuono, Risk factors for hepatitis C virus infection in the Colombian Caribbean coast: A case-control study , Biomedica: Vol. 36 No. 4 (2016)
- Álvaro A. Faccini-Martínez, Elkin G. Forero-Becerra, Jesús A. Cortés-Vecino, Luis J. Polo-Teran, Jorge H. Jácome, Jimmy J. Vargas, Gustavo Valbuena, Marylin Hidalgo, Probable case of flea-borne spotted fever (Rickettsia felis) , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Claudia Lucía Colorado, Guillermo Sánchez, Martha Inírida Guerrero, Clara Inés León, Reliability and agreement of two smear reading scales for classification and monitoring of multidrug therapy in leprosy patients , Biomedica: Vol. 31 No. 3 (2011)
- Juan Gabriel Piñeros, Margarita Arboleda, Juan Camilo Jaramillo, Silvia Blair, Report of five cases of severe neonatal Plasmodium vivax malaria in Urabá, Colombia , Biomedica: Vol. 28 No. 4 (2008)
- Raúl Murillo, Ricardo Cendales, Carolina Wiesner, Marion Piñeros, Sandra Tovar, Effectiveness of cytology-based cervical cancer screening in the Colombian health system , Biomedica: Vol. 29 No. 3 (2009)
- Sandra Lorena Girón, Julio César Mateus, Fabián Méndez, Impact of an open waste disposal site on the occurrence of respiratory symptoms and on health care costs of children , Biomedica: Vol. 29 No. 3 (2009)
- Andrés Páez, Gloria Rey, Carlos Agudelo, Alvaro Dulce, Edgar Parra, Hernando Díaz-Granados, Damaris Heredia, Luis Polo, Outbreak of urban rabies transmitted by dogs in Santa Marta, northern Colombia , Biomedica: Vol. 29 No. 3 (2009)
- Patricia Escobar, Katherine Paola Luna, Indira Paola Hernández, César Mauricio Rueda, María Magdalena Zorro, Simon L. Croft, In vitro susceptibility of Trypanosoma cruzi strains from Santander, Colombia, to hexadecylphosphocholine (miltefosine), nifurtimox and benznidazole , Biomedica: Vol. 29 No. 3 (2009)
- Mauricio Beltrán, María Cristina Navas, María Patricia Arbeláez, Jorge Donado, Sergio Jaramillo, Fernando De la Hoz, Cecilia Estrada, Lucía del Pilar Cortés, Amalia de Maldonado, Gloria Rey, Seroprevalence of hepatitis B virus and human immunodeficiency virus infection in a population of multiply-transfused patients in Colombia , Biomedica: Vol. 29 No. 2 (2009)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























