Anti-inflammatory and antioxidant activity of essential amino acid α-ketoacid analogues against renal ischemia–reperfusion damage in Wistar rats
Abstract
Introduction: Essential amino acid α-keto acid analogs are used in the treatment of chronic kidney disease to delay the symptoms of uremia. However, it is unknown whether essential amino acid α-keto acid analogs affect the oxidative stress and the inflammation in acute renal injury such as those produced by ischemia-reperfusion.
Objective: To evaluate the effect of essential amino acid α-keto acid analogs on renal ischemia-reperfusion injury in Wistar rats.
Materials and methods: Rats were divided into 11 groups (n=6/group): Two groups received physiological saline with or without ischemia-reperfusion injury (45 min/24 h), six groups received essential amino acid α-keto acid analogs (400, 800, or 1,200 mg/kg/24 h/7d) with or without ischemia-reperfusion injury (essential amino acid α-keto acid analogs + ischemia-reperfusion), and two groups received allopurinol (50 mg/kg/24 h/7d) with or without ischemia-reperfusion injury. Biochemical markers included creatinine and blood urea nitrogen (BUN), proinflammatory cytokines (IL-1β, IL-6, and TNF-α), renal damage markers (cystatin C, KIM-1, and NGAL), and markers of oxidative stress such as malondialdehyde (MDA) and total antioxidant activity.
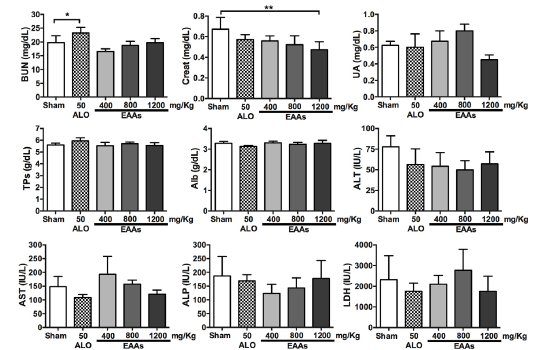
Results: The essential amino acid α-keto acid analog- and allopurinol-treated groups had lower levels of creatinine, BUN, renal damage markers, proinflammatory cytokines, and MDA than their corresponding ischemia-reperfusion groups. These changes were related to the essential amino acid α-keto acid analogs dosage. Total antioxidant activity was lower in essential amino acid α-keto acid analog- and allopurinol-treated groups than in the corresponding ischemia-reperfusion groups.
Conclusions: This is a new report on the nephroprotective effects of essential amino acid α-keto acid analogs against ischemia-reperfusion injury. Essential amino acid α-keto acid analogs decreased the levels of biochemical markers, kidney injury markers, proinflammatory cytokines, and MDA while minimizing total antioxidant consumption.
Downloads
References
Cupisti A, Bolasco P. Keto-analogues and essential aminoacids and other supplements in the conservative management of chronic kidney disease. Panminerva Med. 2017;59:149-56. https://doi.org/10.23736/S0031-0808.16.03288-2
Walser M, Coulter AW, Dighe S, Crantz FR. The effect of keto-analogues of essential amino acids in severe chronic uremia. J Clin Invest. 1973;52:678-90. https://doi.org/10.1172/JCI107229
Jiang Z, Zhang X, Yang L, Li Z, Qin W. Effect of restricted protein diet supplemented with keto analogues in chronic kidney disease: A systematic review and meta-analysis. Int Urol Nephrol. 2016;48:409-18. https://doi.org/10.1007/s11255-015-1170-2
Jefferson JA, Thurman JM. Pathophysiology and etiology of acute kidney injury. Philadelphia: Elsevier; 2015. p. 802-17.
Liu Y. Cellular and molecular mechanisms of renal fibrosis. Nat Rev Nephrol. 2011;7:684-96. https://doi.org/10.1038/nrneph.2011.149
Salvadori M, Rosso G, Bertoni E. Update on ischemia-reperfusion injury in kidney transplantation: Pathogenesis and treatment. World J Transplant. 2015;5:52-67. https://doi.org/10.5500/wjt.v5.i2.52
Wei Q, Dong Z. Mouse model of ischemic acute kidney injury: Technical notes and tricks. Am J Physiol Renal Physiol. 2012;303:F1487-94. https://doi.org/10.1152/ajprenal.00352.2012
Prieto-Moure B, Llori-Carsi JM, Belda-Antoli M, Toledo-Pereyra LH, Cejalvo-Lapeña D. Allopurinol protective effect of renal ischemia by downregulating TNF-α, IL-1β, and IL-6 response. J Invest Surg. 2017;30:143-51. https://doi.org/10.1080/08941939.2016.1230658
Kanno M, Nakayama M, Zhu WJ, Hayashi Y, Kazama JJ. Rosuvastatin pretreatment suppresses distant organ injury following unilateral renal ischemia-reperfusion in hypertensive Dahl saltsensitive rats. Nephrology (Carlton). 2018;23:1046-54 https://doi.org/10.1111/nep.13169
Ziypak T, Halici Z, Alkan E, Akpinar E, Polat B, Adanur S, et al. Renoprotective effect of aliskiren on renal ischemia/reperfusion injury in rats: Electron microscopy and molecular study. Ren Fail. 2015;37:343-54. https://doi.org/10.3109/0886022X.2014.991327
Korkmaz A, Kolankaya D. Protective effect of rutin on the ischemia/reperfusion induced damage in rat kidney. J Surg Res. 2010;164:309-15. https://doi.org/10.1016/j.jss.2009.03.022
Collins AJ, Couser WG, Dirks JH, Kopple JD, Reiser T, Riella MC, et al. World Kidney Day: An idea whose time has come. Kidney Int. 2006;69:781-2. https://doi.org/10.1038/sj.ki.5000250
Eddy AA. Molecular insights into renal interstitial fibrosis. J Am Soc Nephrol. 1996;7:2495-508.
Garneata L, Mircescu G. Effect of low-protein diet supplemented with keto acids on progression of chronic kidney disease. J Ren Nutr. 2013;23:210-3. https://doi.org/10.1053/j.jrn.2013.01.030
Gao X, Wu J, Dong Z, Hua C, Hu H, Mei C. A low-protein diet supplemented with ketoacids plays a more protective role against oxidative stress of rat kidney tissue with 5/6 nephrectomy than a low-protein diet alone. Br J Nutr. 2010;103:608-16. https://doi.org/10.1017/S0007114509992108
Peng H, Mao Y, Fu X, Feng Z, Xu J. Comparison of biomarkers in rat renal ischemia-reperfusion injury. Int J Clin Exp Med. 2015;8:7577-84.
Li HY, Hou FF, Zhang X, Chen PY, Liu SX, Feng JX, et al. Advanced oxidation protein products accelerate renal fibrosis in a remnant kidney model. J Am Soc Nephrol. 2007;18:528-38. https://doi.org/10.1681/ASN.2006070781
Chen W, Guo ZY, Wu H, Sun LJ, Cai LL, Xu HY. Effects of low-protein diet plus alpha-keto acid on micro-inflammation and the relationship between micro-inflammation and nutritional status in patients performing continuous ambulatory peritoneal dialysis: A randomized controlled trial. Chin J Integr Med. 2008;6:473-7. https://doi.org/10.3736/jcim20080508
Zhang Y, Huang J, Yang M, Gu L, Ji J, Wang W, et al. Effect of a low-protein diet supplemented with keto-acids on autophagy and inflammation in 5/6 nephrectomized rats. Biosci Rep. 2015;35:e00263. https://doi.org/10.1042/BSR20150069
Liu D, Wu M, Li L, Gao X, Yang B, Mei S, et al. Low-protein diet supplemented with ketoacids delays the progression of diabetic nephropathy by inhibiting oxidative stress in the KKAy mice model. Br J Nutr. 2018;119:22-9. https://doi.org/10.1017/S0007114517003208
Rabadi MM, Ghaly T, Goligorksy MS, Ratliff BB. HMGB1 in renal ischemic injury. Am J Physiol Renal Physiol. 2012;303:F873-F85. https://doi.org/10.1152/ajprenal.00092.2012
Chung KY, Park JJ, Kim YS. The role of high-mobility group box-1 in renal ischemia and reperfusion injury and the effect of ethyl pyruvate. Transplant Proc. 2008;40:2136-8. https://doi.org/10.1016/j.transproceed.2008.06.040
Mori da Cunha MG, Zia S, Beckmann DV, Carlon MS, Arcolino FO, Albersen M, et al. Vascular endothelial growth factor up-regulation in human amniotic fluid stem cell enhances nephroprotection after ischemia-reperfusion injury in the rat. Crit Care Med. 2017;45:e86-e96. https://doi.org/10.1097/CCM.0000000000002020
Milovanova L, Fomin V, Moiseev S, Taranova M, Milovanov Y, Lysenko-Kozlovskaya L, et al. Effect of essential amino acid кetoanalogues and protein restriction diet on morphogenetic proteins (FGF-23 and Кlotho) in 3b-4 stages chronic кidney disease patients: a randomized pilot study. Clin Exp Nephrol. 2018;6:1351-9. https://doi.org/10.1007/s10157-018-1591-1
Some similar items:
- Vanihamín Domínguez, Itzen Aguiñiga, Leticia Moreno, Beatriz Torres, Edelmiro Santiago-Osorio, Sodium caseinate increases the number of B lymphocytes in mouse , Biomedica: Vol. 37 No. 4 (2017)
- Luis A. Franco, Germán E. Matiz, Jairo Calle, Roberto Pinzón, Luis F. Ospina, Antiinflammatory activity of extracts and fractions obtained from Physalis peruviana L. calyces , Biomedica: Vol. 27 No. 1 (2007)
- Edisson Rodríguez, Aura María Gil-Villa, Daniel Camilo Aguirre-Acevedo, Walter Cardona-Maya, Ángela P. Cadavid, Evaluation of atypical semen parameters in individuals whose couples had a history of early recurrent embryo death: in search for a reference value , Biomedica: Vol. 31 No. 1 (2011)
- Luis Ángel Villar, Rosa Margarita Gélvez, Jairo Antonio Rodríguez, Doris Salgado, Beatriz Parra, Lyda Osorio, Irene Bosch, Biomarkers for the prognosis of severe dengue , Biomedica: Vol. 33 (2013): Suplemento 1, Fiebres hemorrágicas
- Fabián Jaimes, Gisela de la Rosa, Anticoagulation and sepsis: the opportunity for a new use of heparin?. , Biomedica: Vol. 26 No. 1 (2006)
- Luis Alberto Gómez, María Luz Gunturiz, Identification and differential expression of the microphthalmia-associated transcription factor in heart and isolated cardiomyocytes from Guinea pigs: Possible role in hypertrophy and viability , Biomedica: Vol. 34 No. 3 (2014)
- Henry A. Vargas, Martín Rondón, Rodolfo Dennis, Pharmacological treatment and impairment of pulmonary function in patients with type 2 diabetes: a cross-sectional study , Biomedica: Vol. 36 No. 2 (2016)
- Claudia Viviana Barbosa, Carlos Enrique Muskus, Luz Yaneth Orozco, Adriana Pabón, Mutagenicity, genotoxicity and gene expression of Rad51C, Xiap, P53 and Nrf2 induced by antimalarial extracts of plants collected from the middle Vaupés region, Colombia , Biomedica: Vol. 37 No. 3 (2017)
- Manuela Velásquez, Manuel Alejandro Granada, Juan Camilo Galvis, Ángela María Álvarez, Ángela Cadavid, Oxidative stress in endothelial cells induced by the serum of women with different clinical manifestations of the antiphospholipid syndrome , Biomedica: Vol. 39 No. 4 (2019)
- Juana P. Sánchez-Villamil, Carolina Pino-Vélez, Juanita Trejos-Suárez, Néstor Cardona , Dra. Ana, Pedro A. Alfonso, Salivary markers of oxidative stress and periodontal pathogens in patients with periodontitis from Santander, Colombia , Biomedica: Vol. 40 No. Supl. 1 (2020): Mayo, Infecciones en el trópico
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























