Etiological identification of viral agents in acute encephalitis in Guadalajara, México, 2011-2015
Abstract
Introduction: Viral encephalitis is a well-known inflammatory process associated with neurological dysfunction that might derive into severe brain damage or a fatal outcome. In México there is no epidemiological data that describes the prevalence of viral agents responsible for acute encephalitis.
Objective: To identify the main viral agents by real time PCR involved in acute encephalitis in Mexico.
Materials and methods: We obtained cerebral spinal fluid (CSF) samples from all patients with suspected viral encephalitis admitted to the emergency service of the Hospital Civil de Guadalajara “Fray Antonio Alcalde”. To identify pathogens, we performed nucleic acid extraction using real-time PCR and RT-PCR.
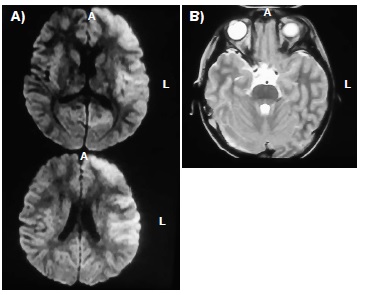
Results: Sixty-six patients were diagnosed with acute encephalitis from 2011 to 2014. A definitive viral etiological diagnosis was established in 16 patients (24%); the main causative agents were enteroviruses in 50% of the 16 positive samples, followed by herpes simplex virus (37%) and cytomegaloviruses (12.5%). Patients with encephalitis were predominantly male (63.3%) and a seasonal predominance was observed during autumn (37.5%). The main clinical characteristics in the acute encephalitis phase were fever (48.45) and cephalea (36.3), followed by seizures, disorientation, and muscular weakness (30.3%). Kerning sign was present in two cases (3%) and other two cases presented Brudzinski’s sign (3%).
Conclusions: CSF PCR is a suitable diagnostic technique for the identification of viral encephalitis caused by viral infections that allows an appropriate antiviral therapeutic treatment.
Downloads
References
Hamid JS, Meaney C, Crowcroft NS, Granerod J, Beyene J, UK Health Protection Agency Aetiology of Encephalitis Study Group. Potential risk factors associated with human encephalitis: Application of canonical correlation analysis. BMC Med Res Methodol. 2011;11:120. https://doi.org/10.1186/1471-2288-11-120
Tunkel AR, Glaser CA, Bloch KC, Sejvar JJ, Marra CM, Roos KL, et al. The management of encephalitis: Clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2008;47:303-27. https://doi.org/10.1086/589747
Stahl JP, Mailles A, Dacheux L, Morand P. Epidemiology of viral encephalitis in 2011. Med Mal Infect. 2011;41:453-64. https://doi.org/10.1016/j.medmal.2011.05.015
Somand D, Meuer W. Central nervous system infections. Emerg Med Clin North Am. 2009;27:89-100. https://doi.org/10.1016/j.emc.2008.07.004
Mailles A, Stahl JP. Infectious encephalitis in France in 2007: A national prospective study. Clini Infect Dis. 2009;49:1838-47. https://doi.org/10.1086/648419
Arroyo HA, Bologna R. Viral encephalitis. Rev Neurol. 1997;142:912-9.
Mace SE. Central nervous system infections as a cause of an altered mental status? What is the pathogen growing in your central nervous system? Emerg Med Clin North Am. 2010;28:535-70. https://doi.org/10.1016/j.emc.2010.03.002
Cho TA, Mckendall RR. Clinical approach to the syndromes of viral encephalitis, myelitis, and meningitis. Handb Clin Neurol. 2014;123:89-121. https://doi.org/10.1016/B978-0-444-53488-0.00004-3
Watzinger F, Suda M, Preuner S, Baumgartinger R, Ebner K, Baskova L, et al. Real-time quantitative PCR assays for detection and monitoring of pathogenic human viruses in immunosuppressed pediatric patients. J Clin Microbiol. 2004;42:5189-98. https://doi.org/10.1128/JCM.42.11.5189–5198.2004
Debiasi RL, Tyler KL. Molecular methods for diagnosis of viral encephalitis. Clin Microbiol Rev. 2004;17:903-25. https://doi.org/10.1128/CMR.17.4.903-925.2004
Rodríguez SR, Gómez BD, Pallansch M. Brote epidémico de meningitis viral causado por virus Echo tipo 30. Bol Med Hosp Infant Mex. 1992;49:412-5.
Rodríguez ML, Rodríguez DR, Blitvich BJ, López MA, Fernández-Salas I, Jiménez JR, et al. Serologic surveillance for West Nile virus and other flaviviruses in febrile patients, encephalitic patients, and asymptomatic blood donors in northern México. Vector Borne Zoonotic Dis. 2010;10:151-7. https://doi.org/10.1089/vbz.2008.0203
Velásquez-Pérez L, Ramírez-Crescencio MA. Neurological disease surveillance and mandatory reporting: A trend and outcome of the national neurological institute of health in México city from 2005 to 2011. Gac Med Mex. 2014;150:540-51.
Stankiewicz PI, Piotrowska A. Meningitis and encephalitis in Poland in 2013. Przegl Epidemiol. 2015;69:229-34.
Stahl JP, Mailles A. What is new about epidemiology of acute infectious encephalitis? Curr Opin Neurol. 2014;27:337-41. https://doi.org/10.1097/WCO.0000000000000097
Fitch MT, Abrahamian FM, Moran GJ, Talan DA. Emergency department management of meningitis and encephalitis. Infect Dis Clin of North Am. 2008;22:33-52. https://doi.org/10.1016/j.idc.2007.10.001
Li W, Zhang X, Chen X, Cheng YP, Wu YD, Shu Q. Epidemiology of childhood enterovirus infections in Hangzhou, China. Virol J. 2015;12:58. https://doi.org/10.1186/s12985-015-0294-4
Flóres-González JC, Jordán-García I, Turón-Viñas E, Montero-Valladares C, Téllez-González C, Fernández-Carrión F, et al. Etiología, presentación clínica y evolución neurológica de las encefalitis víricas graves en la edad pediátrica (estudio ECOVE). Rev Neurol. 2015;61:7-13.
Fica A, Pérez C, Reyes P, Gallardo S, Calvo X, Salinas AM. Serie clínica de 15 casos confirmados por reacción de polimerasa en cadena. Rev Chil Infect. 2005;22:38-46. https://doi.org/10.4067/S0716-10182005000100005
Hebant B, Miret N, Bouwyn JP, Delafosse E, Lefaucheur R. Absence of pleocytosis in cerebrospinal fluid does not exclude herpes simplex virus encephalitis in elderly adults. J Am Geriatr Soc. 2015;63:1278-9. https://doi.org/10.1111/jgs.13493
Patel B, Bhatt GC, Kushwaha KP, Gore MM. Japanese encephalitis presenting without cerebrospinal fluid pleocytosis. Pediatr Infect Dis J. 2015;34:1416. https://doi.org/10.1097/INF.0000000000000911
Doughty CT, Yawetz S, Lyons J. Emerging causes of arbovirus encephalitis in North America: Powassan, Chikungunya, and Zika viruses. Curr Neurol Neurosci Rep. 2017;17:12. https://doi.org/10.1007/s11910-017-0724-3
Acevedo N, Waggoner J, Rodríguez M, Rivera L, Landivar J, Pinsky B, et al. Zika virus, Chikungunya virus, and dengue virus in cerebrospinal fluid from adults with neurological manifestations, Guayaquil, Ecuador. Front Microbiol. 2017;8:42. https://oi.org/10.3389/fmicb.2017.00042
Mailles A, Stahl JP, Bloch KC. Update and new insights in encephalitis. Clin Microbiol Infect. 2017;23: 607-13. https://doi.org/10.1016/j.cmi.2017.05.002
Some similar items:
- Marco Torres-Castro, Biol. Naomi Cuevas, Dra. Silvia Hernández, IBQ. Henry Noh, Biol. Erendira Estrella, M. en C. Belén Herrera, Dr. Alonso Panti, Dr. Etienne Waleckx, M. en C. Javier Sosa, Dr. Ronald Peláez, Natural infection with Trypanosoma cruzi in bats captured in Campeche and Yucatán, México , Biomedica: Vol. 41 No. Supl. 1 (2021): Mayo, Parasitología médica
- Luis Guillermo Uribe, María Alejandra Pérez, Camilo Andrés Lara, Natalia Rueda, Javier Augusto Hernández, Wernicke-Korsakoff syndrome secondary to cytomegalovirus encephalitis: A case report , Biomedica: Vol. 37 No. 4 (2017)
- Sonia P. Bohórquez, Juliana Díaz, Claudia M. Rincón, Marcela Estupiñán, Mauricio Chaparro, Ana María Low-Calle, Jaime E. Castellanos, Shedding of HSV-1, HSV-2, CMV, and EBV in the saliva of hematopoietic stem cell transplant recipients at Fundación HOMI - Hospital de la Misericordia, Bogotá, D.C. , Biomedica: Vol. 36 (2016): Suplemento 2, Enfermedades virales
- Ernesto Pérez-Sánchez, Raúl Montiel-Cruz, Eréndira Romero-Domínguez, Griselda Pascacio-Bermúdez, Arturo Báez-Hernández, Guadalupe Díaz del Castillo-Flores, Fabián Correa-Morales, Gonzalo Vázquez-Prokopec, Pablo Manrique-Saide, Azael Che-Mendoza, Gabriela Meneses-Ruiz , Irma López-Martínez, María Jesús Sánchez, Seroprevalence of Trypanosoma cruzi among children from Veracruz, Mexico: Epidemiological baseline for a control model based on Chagas disease active transmission , Biomedica: Vol. 44 No. 1 (2024)
- Mercedes Salcedo-Cifuentes, Jesús Cabrera, Yesid Cuesta-Astroz, Edwin Carrascal, Yoshito Eizuru, Martha C. Domínguez, Adalberto Sánchez, Felipe García-Vallejo, Clonal expansion and genomic characterization of the human T-cell lymphotropic virus type I during the integration process in adult T-cell leukemia/lymphoma , Biomedica: Vol. 29 No. 2 (2009)
- Martine Bonnaure-Mallet, Paula Juliana Pérez-Chaparro, Patrice Gracieux, Vincent Meuric, Zohreh Tamanai-Shacoori, Jaime Eduardo Castellanos, Distribution of Porphyromonas gingivalis fimA genotypes in isolates from subgingival plaque and blood sample during bacteremia , Biomedica: Vol. 29 No. 2 (2009)
- Diego Fernando Zea, Martín Prager, Roger Adrian Figueroa, María Consuelo Miranda, Mucosal complication of cutaneous leishmaniasis , Biomedica: Vol. 29 No. 1 (2009)
- Ana Margarita Montalvo, Lianet Monzote, Jorge Fraga, Ivón Montano, Carlos Muskus, Marcel Marín, Simonne De Donck, Iván Darío Vélez, Jean Claude Dujardin, PCR-RFLP and RAPD for typing neotropical Leishmania , Biomedica: Vol. 28 No. 4 (2008)
- Concepción Judith Puerta, Johana María Guevara, Paula Ximena Pavía, Marleny Montilla, Rubén Santiago Nicholls, Edgar Parra, Yuli Katherine Barrera, Evaluation of TcH2AF-R and S35-S36 primers in PCR tests for the detection of Trypanosoma cruzi in mouse cardiac tissue , Biomedica: Vol. 28 No. 4 (2008)
- María Cristina Ferro, Victor Alberto Olano, Martha Ahumada, Scott Weaver, Mosquitos (Diptera: Culicidae) in the small village where a human case of Venezuelan equine encephalitis was recorded , Biomedica: Vol. 28 No. 2 (2008)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























