An outbreak of Salmonella enterica subsp. enterica serovar Give associated with foodborne illness in the department of Vichada, Colombia, 2015
Abstract
Introduction: Salmonella enterica subsp. enterica serovar Give is found in ruminants, pigs, poultry, and aquatic environments, but rarely in humans. In Colombia, this serotype was ranked 11th. in the laboratory surveillance of acute diarrheal disease between 2000 and 2013.
Objective: To characterize phenotypic and genotypic isolates of Salmonella related to an outbreak of foodborne Illness in the department Vichada in the fifth epidemiological week of 2015.
Materials and methods: Following the Instituto Nacional de Salud method, we tested 37 fecal samples for Salmonella spp. while the sample of canned sardines was processed according to the ISO 6579:2002 Cor.1:2004 standard. The isolates were confirmed by serology and/or real-time PCR, antimicrobial susceptibility tests, and pulsed-field gel electrophoresis with the XbaI and BlnI enzymes.
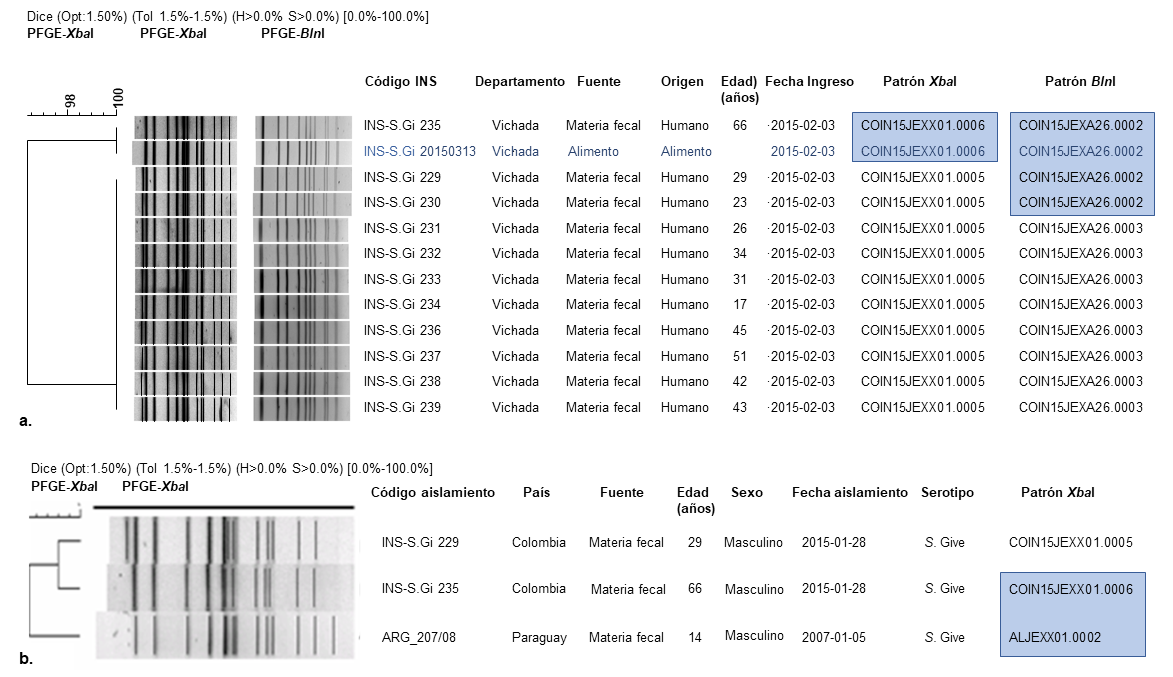
Results: All human isolates (11) and that from food (1) were identified as S. Give. The food isolate exhibited tetracycline resistance. PFGE analysis with XbaI grouped ten isolates from samples of human origin in pattern COIN15JEXX01.0005 and the remaining isolates in COIN15JEXX01.0006 with 96.3% similarity. All isolates were confirmed with the BlnI enzyme, and four (three human isolates and the one from food) were matched to the pattern COIN15JEXA26.002 with 95.65% similarity.
Conclusion: Our study confirmed that canned sardines were related to the transmission of S. Give in the outbreak, which is the third one caused by this serotype in Colombia.
Downloads
References
World Health Organization. WHO estimates of the global burden of foodborne diseases: Foodborne disease burden epidemiology reference group 2007-2015. Geneva: WHO; 2015. p. 268.
Guerrero Montilla JA. Informe final del evento enfermedades transmitidas por alimentos, Colombia, 2015. Bogotá: Instituto Nacional de Salud; 2016. p. 17. Fecha de consulta: 5 de septiembre de 2019. Disponible en: https://www.ins.gov.co/buscador-eventos/Informesdeevento/ETA%202015.pdf
Girardin F, Mezger N, Hächler H, Bovier PA. Salmonella serovar Give: An unusual pathogen causing splenic abscess. Eur J Clin Microbiol Infect Dis. 2006;25:272-4. https://doi.org/10.1007/s10096-006-0122-2
Usera MA, Aladueña A, Díez R, Cerdán P, Gutiérrez R, Echeita A. Análisis de los serotipos de de Salmonella sp. aisladas de muestras no humanas en 1996 en España. Boletín Epidemiológico Semanal. España: Instituto de Salud Carlos III; 1997. p. 69-80. Fecha de consulta: 11 de febrero de 2019. Disponible en: http://www.adiveter.com/ftp_public/articulo450.pdf
Jourdan N, Hello S Le, Delmas G, Clouzeau J, Manteau C, Désaubliaux B, et al. Nationwide outbreak of Salmonella enterica serotype Give infections in infants in France, linked to infant milk formula, September 2008. Eurosurveillance. 2008;13:1-2. https://www.eurosurveillance.org/docserver/fulltext/eurosurveillance/13/39/art18994- en.pdf? expires=1613766271&id=id&accname=guest&checksum=E754A4D07E0DF47C1AF 2AAEE2B21FB0E
Borriello G, Lucibelli MG, Pesciaroli M, Carullo MR, Graziani C, Ammendola S, et al. Diversity of Salmonella spp. serovars isolated from the intestines of water buffalo calves with gastroenteritis. BMC Vet Res. 2012;8:1-9. https://doi.org/10.1186/1746-6148-8-201
Maurer JJ, Martín G, Hernández S, Cheng Y, Gerner-Smidt P, Hise KB, et al. Diversity and persistence of Salmonella enterica strains in rural landscapes in the southeastern United States. PLoS ONE. 2015;10:1-19. https://doi.org/10.1371/journal.pone.0128937
Instituto Nacional de Salud. Características de los aislamientos de Salmonella spp. Colombia, Resultados de la vigilancia 2000-2013. p. 1-31. Fecha de consulta: 17 de noviembre de 2019. Disponible en: https://www.ins.gov.co/buscador-eventos/Informacin%20de%20laboratorio/Informe%20Vigilancia%20por%20laboratorio%20de%20Salmonella%20spp%202000-2013.pdf
International Standard ISO. Microbiology of food and animal feeding stuffs - Horizontal method for the detection of Salmonella spp. (ISO 6579:2002). Fecha de consulta: 15 de diciembre de 2016. Disponible en: https://www.iso.org/standard/40377.html
Muñoz N, Díaz-Osorio M, Moreno J, Sánchez-Jiménez M, Cardona-Castro N. Development and evaluation of a multiplex real-time polymerase chain reaction procedure to clinically type prevalent Salmonella enterica serovars. J Mol Diagnostics. 2010;12:220-5. https://doi.org/10.2353/jmoldx.2010.090036
Grimont PA, Weill F-X. Antigenic Formulae of the Salmonella Serovars. 9th edition. WHO Collaborating Centre for Reference and Research on Salmonella, editor. Paris: Institute Pasteur; 2007. p. 1-166.
Caffer MI, Terragno R. Manual de procedimientos para la caracterización de Salmonella. Buenos Aires: Ministerio de Salud; 2001. p. 1-37. Fecha de consulta: 11 de febrero de 2019. Disponible en: https://docplayer.es/12601568-Manual-de-procedimientos-para-lacaracterizacion-de-salmonella.html
Realpe M, Montaño LA. Manual de procedimientos para el diagnóstico bacteriológico de enfermedad diarreica bacteriana aguda, identificación de Salmonella spp., Shigella sp. y Vibrio cholerae. Bogotá: Instituto Nacional de Salud; 2015.
Centers for Disease Control and Prevention. Standard Operating Procedure for PulseNet PFGE of Escherichia coli O157:H7, Escherichia coli non-O157 (STEC), Salmonella serotypes, Shigella sonnei and Shigella flexneri. Internet. CDC PulseNet. PNL05 Last Updated December 2017. p. 1-16. Fecha de consulta: 17 de julio de 2019. Disponible en: https://www.cdc.gov/pulsenet/PDF/ecoli-shigella-salmonella-pfge-protocol-508c.pdf
Roy R, Higgins R, Fortin M, Tardif S. Salmonella Give infection in 2 dairy herds. Can Vet J. 2001;42:468-70.
Jansen A, Frank C, Prager R, Oppermann H, Stark K. Bundesweiter Ausbruch durch Salmonella Give in Deutschland im Jahr 2004. Nation-wide Outbreak of Salmonella Give in Germany, 2004. Z Gastroenterol. 2005;43:707-13. https://doi.org/10.1055/s-2005-858256
Tenover FC, Arbeit RD, Goering R V, Mickelsen PA, Murray BE, Persing DH, et al. Interpreting chromosomal DNA restriction patterns produced by pulsed- field gel electrophoresis: Criteria for bacterial strain typing. J Clin Microbiol. 1995;33:2233-9. https://doi.org/10.1128/JCM.33.9.2233-2239.1995
McClelland M, Jones R, Patel Y, Nelson M. Restriction endonucleases for pulsed field mapping of bacterial genomes. Nucleic Acids Res. 1987;15:5985-6005. https://doi.org/10.1093/nar/15.15.5985
Ribot EM, Fair MA, Gautom R, Cameron DN, Hunter SB, Swaminathan B, et al. Standardization of pulsed-field gel electrophoresis protocols for the subtyping of Escherichia coli O157:H7, Salmonella, and Shigella for PulseNet. Foodborne Pathog Dis. 2006;3:59-67. https://doi.org/10.1089/fpd.2006.3.59
Zheng J, Keys CE, Zhao S, Ahmed R, Meng J, Brown EW. Simultaneous analysis of multiple enzymes increases accuracy of pulsed-field gel electrophoresis in assigning genetic relationships among homogeneous Salmonella strains. J Clin Microbiol. 2011;49:85-94. https://doi.org/10.1128/JCM.00120-10
Goering RV, Tenover FC. Epidemiological interpretation of chromosomal macrorestriction fragment patterns analyzed by pulsed-field gel electrophoresis. J Clin Microbiol. 1997;35:2432-3. https://doi.org/10.1128/JCM.35.9.2432-2433.1997
Galitsopoulou A, Georgantelis D, Kontominas M. The influence of industrial-scale canning on cadmium and lead levels in sardines and anchovies from commercial fishing centres of the Mediterranean Sea. Food Addit Contam Part B Surveill. 2012;5:75-81. https://doi.org/10.1080/19393210.2012.658582
Mol S. Levels of heavy metals in canned bonito, sardines, and mackerel produced in Turkey. Biol Trace Elem Res. 2011;143:974-82. https://doi.org/10.1007/s12011-010-8909-5
Shiber JG. Arsenic, cadmium, lead and mercury in canned sardines commercially available in eastern Kentucky, USA. Mar Pollut Bull. 2011;62:66-72. https://doi.org/10.1016/j.marpolbul.2010.09.008
Castillo A, Mercado I, Lucía LM, Martínez-Ruiz Y, Ponce De León J, Murano EA, et al. Salmonella contamination during production of cantaloupe: A binational study. J Food Prot. 2004;67:713-20. https://doi.org/10.4315/0362-028X-67.4.713
Higgins R, Désilets A, Cantin M, Messier S, Khakhria R, Ismaïl J, et al. Outbreak of Salmonella Give in the Province of Quebec. Can Vet J. 1997;38:780-1.
Jiménez M, Martínez-Urtaza J, Rodríguez-Álvarez MX, León-Felix J, Chaidez C. Prevalence and genetic diversity of Salmonella spp. in a river in a tropical environment in México. J Water Health. 2014;12:874-84. https://doi.org/10.2166/wh.2014.051
Tracogna MF, Lösch LS, Alonso JM, Merino LA. Detection and characterization of Salmonella spp. in recreational aquatic environments in the Northeast of Argentina. Rev Ambient Agua. 2013;8:18-26. https://doi.org/10.4136/ambi-agua.1145
Traoré O, Nyholm O, Siitonen A, Bonkoungou IJO, Traoré AS, Barro N, et al. Prevalence and diversity of Salmonella enterica in water, fish and lettuce in Ouagadougou, Burkina Faso. BMC Microbiol. 2015;15:1-7. https://doi.org/10.1186/s12866-015-0484-7
Fletcher S. Understanding the contribution of environmental factors in the spread of antimicrobial resistance. Environ Health Prev Med. 2015;20:243-52. https://doi.org/10.1007/s12199-015-0468-0
Molina A, Granados-Chinchilla F, Jiménez M, Acuña-Calvo MT, Alfaro M, Chavarría G. Vigilance for Salmonella in feedstuffs available in Costa Rica: Prevalence, serotyping and tetracycline resistance of isolates obtained from 2009 to 2014. Foodborne Pathog Dis. 2016;13:119-27. https://doi.org/10.1089/fpd.2015.2050
González F, Araque M. Association of transferable quinolone resistance determinant qnrB19 with extended-spectrum β-lactamases in Salmonella Give and Salmonella Heidelberg in Venezuela. Int J Microbiol. 2013;2013:6. https://doi.org/10.1155/2013/628185
Chinen I, Campos J, Dorji T, Pérez-Gutiérrez E. PulseNet Latin America and the Caribbean Network: Present and future. Foodborne Pathog Dis. 2019;16:489-97. https://doi.org/10.1089/fpd.2018.2587
Some similar items:
- Elsa De La Cadena, Christian José Pallares, Juan Carlos García-Betancur, Jessica A. Porras, María Virginia Villegas, Update of antimicrobial resistance in level III and IV health institutions in Colombia between January 2018 and December 2021 , Biomedica: Vol. 43 No. 4 (2023)
- Alexandra Hurtado-Ortiz, José Moreno-Montoya , Franklyn E. Prieto-Alvarado , Álvaro J. Idrovo, Benchmarking of public health surveillance of COVID-19 in Colombia: First semester , Biomedica: Vol. 40 No. Supl. 2 (2020): SARS-CoV-2 y COVID-19
- Edgar F. Manrique-Hernández, José Moreno-Montoya, Alexandra Hurtado-Ortiz, Franklyn E. Prieto-Alvarado, Álvaro J. Idrovo, Performance of the Colombian surveillance system during the COVID-19 pandemic: A rapid evaluation of the first 50 days , Biomedica: Vol. 40 No. Supl. 2 (2020): SARS-CoV-2 y COVID-19
- Jaime E. Bernal, Martha Lucía Tamayo , Ignacio Briceño , Escilda Benavides , Newborn screening in Colombia: The experience of a private program in Bogotá , Biomedica: Vol. 44 No. 1 (2024)
- Raúl Murillo, Ricardo Cendales, Carolina Wiesner, Marion Piñeros, Sandra Tovar, Effectiveness of cytology-based cervical cancer screening in the Colombian health system , Biomedica: Vol. 29 No. 3 (2009)
- Sandra Lorena Girón, Julio César Mateus, Fabián Méndez, Impact of an open waste disposal site on the occurrence of respiratory symptoms and on health care costs of children , Biomedica: Vol. 29 No. 3 (2009)
- José Joaquín Carvajal, Ligia Inés Moncada, Mauricio Humberto Rodríguez, Ligia del Pilar Pérez, Víctor Alberto Olano, Characterization of Aedes albopictus (Skuse, 1894) (Diptera:Culicidae) larval habitats near the Amazon River in Colombia , Biomedica: Vol. 29 No. 3 (2009)
- Andrés Páez, Gloria Rey, Carlos Agudelo, Alvaro Dulce, Edgar Parra, Hernando Díaz-Granados, Damaris Heredia, Luis Polo, Outbreak of urban rabies transmitted by dogs in Santa Marta, northern Colombia , Biomedica: Vol. 29 No. 3 (2009)
- Patricia Escobar, Katherine Paola Luna, Indira Paola Hernández, César Mauricio Rueda, María Magdalena Zorro, Simon L. Croft, In vitro susceptibility of Trypanosoma cruzi strains from Santander, Colombia, to hexadecylphosphocholine (miltefosine), nifurtimox and benznidazole , Biomedica: Vol. 29 No. 3 (2009)
- Mauricio Beltrán, María Cristina Navas, María Patricia Arbeláez, Jorge Donado, Sergio Jaramillo, Fernando De la Hoz, Cecilia Estrada, Lucía del Pilar Cortés, Amalia de Maldonado, Gloria Rey, Seroprevalence of hepatitis B virus and human immunodeficiency virus infection in a population of multiply-transfused patients in Colombia , Biomedica: Vol. 29 No. 2 (2009)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























