Detection of Toxoplasma gondii in water for human consumption from water wells (jagüeyes) in the rural area of the municipality of Sincelejo
Abstract
Introduction: Toxoplasmosis is an orally-transmitted zoonosis that may appear after consuming food contaminated with any infective form of Toxoplasma gondii. Its transmission by water has been reported in several countries including Colombia. The rural population of Sincelejo could be at risk of contracting toxoplasmosis through this route given that they lack potable water.
Objective: To evaluate T. gondii contamination in water for human consumption from water wells (jagüeyes) in the rural area of Sincelejo and establish its relationship with different social determinants of health in the study area.
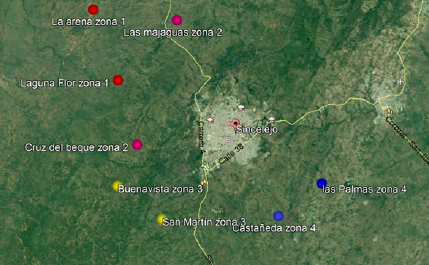
Materials and methods: Using nested PCR we evaluated 96 water samples obtained from 48 farms located in eight rural townships in Sincelejo. We took two samples in each farm: one of raw water from water wells and the other intended for direct consumption. We conducted a survey on each farm to collect information on the physical characteristics of dwellings, the presence of cats, and the availability and uses of water. Statistical relationships were evaluated through Fisher tests.
Results: Of the 96 samples analyzed, 13 were contaminated with T. gondii (13.5%): Nine corresponded to raw water and four to water for direct consumption. No statistical association was found between the positive samples and the social determinants of health under evaluation (p>0.05).
Conclusion: The rural population of Sincelejo could be at risk of contracting toxoplasmosis through the use and/or consumption of water from its water wells. The contamination of these water bodies by T. gondii may be influenced by unstudied social determinants.
Downloads
References
Abu-Madi MA, Al-Molawi N, Behnke JM. Seroprevalence and epidemiological correlates of Toxoplasma gondii infections among patients referred for hospital-based serological testing in Doha, Qatar. Parasit Vectors. 2008;1:1-9. https://doi.org/10.1186/1756-3305-1-39
Carrada TB. Toxoplasmosis: parasitosis reemergente del nuevo milenio. Rev Mex Patol Clin. 2005;52:151-62.
Sánchez R, Cobos D, Miranda A, Goya Y, Miranda A. Aspectos básicos sobre la patogenia, respuesta inmune y bioseguridad en el trabajo con el Toxoplasma gondii. Correo Científico Médico. 2012;16:1-16.
Simon A, Bigras M, Rousseau AN, Dubey JP, Ogden NH. Spatiotemporal dynamics of Toxoplasma gondii infection in Canadian lynx (Lynx canadensis) in western Québec,
Canada. J Wildl Dis. 2013;49:39-48. https://doi.org/10.7589/2012-02-048
Gómez F. Estudio sobre la toxoplasmosis en Andorra y el Alt Urgell (tesis). Barcelona: Universidad de Barcelona; 2004.
Alvarado F. Toxoplasmosis en el Inmunosuprimido. Rev Salud Pública. 2002;4:31-4.
Lizarazo J, Castro F, De Arco M, Chaves C, Peña Y. Infecciones oportunistas del sistema nervioso central en pacientes con VIH atendidos en el Hospital Universitario Erasmo Meoz, Cúcuta, 1995-2005. Infectio. 2006;10:226-31.
Ávila G, González G. Algunas manifestaciones neurológicas del síndrome de inmunodeficiencia adquirida (sida) en pacientes del hospital universitario Hernando Moncaleano Perdomo de Neiva, 2001 -2004. Acta Biológica Colombiana. 2007;23:90-4.
Campo DM, Discuviche MA, Blanco PJ, Montero YM, Orozco KE, Assia YM. Detección de Toxoplasma gondii por amplificación del gen B1 en carnes de consumo humano. Infectio. 2014;18:93-9. https://doi.org/10.1016/j.infect.2014.05.001
Giraldo M. Toxoplasmosis. Medicina & Laboratorio. 2008;14:359-75.
Cortés J, Gómez J, Silva P, Arévalo L, Arévalo I, Álvarez M, et al. Guía de atención integral para la prevención, detección temprana y tratamiento de las complicaciones del embarazo, parto y puerperio: sección toxoplasmosis en el embarazo. Infectio. 2012;16:230-46. https://doi.org/10.1016/S0123-9392(12)70018-8
Pino LE, Salinas JE. Descripción de un brote epidémico de toxoplasmosis aguda en pacientes inmunocompetentes miembros de las fuerzas militares de Colombia durante
operaciones de selva. Infectio. 2009;13:83-91. https://doi.org/10.1016/S0123-9392(09)70729-5
Arcia C. Detección molecular de comunidades microbianas en agua para el consumo humano en el municipio de sincelejo, Sucre- Colombia (tesis). Sincelejo: Universidad de Córdoba; 2016.
Sánchez C. Detección y caracterización molecular de los parásitos de interés en salud pública: Giardia duodenalis, Cryptosporidium spp., Cyclospora cayetanensis, Toxoplasma gondii y Entamoeba histolytica, en agua cruda y tratada de cuatro plantas potabilizadoras del departamento de Nariño (Colombia) (tesis). Bogotá: Universidad Nacional de Colombia; 2017.
Triviño J, Lora F, Zuluaga J, Gómez, J. Detection by PCR of pathogenic protozoa in raw and drinkable water samples in Colombia. Parasitol Res. 2016;115:1789-97. https://doi.org/10.1007/s00436-016-4917-5
Luna JC, Zamora A, Hernández-Arango N, Muñoz-Sánchez D, Pinzón MI, Cortés-Vecino JA, et al. Food safety assessment and risk for toxoplasmosis in school restaurants in Armenia, Colombia. Parasitol Res. 2019;118:3449-57. https://doi.org/10.1007/s00436-019-06473-w
Ballut-Dajud G, Monroy-Pineda MC. Los jagüeyes del municipio de Sincelejo, Sucre, Colombia. Rev Colomb Cienc Anim. 2015;7:80-3. https://doi.org/10.24188/recia.v7.n1.2015.426
Sroka J, Wójcik-Fatla A, Dutkiewicz J. Occurrence of Toxoplasma gondii in water from wells located on farms. Ann Agric Environ Med. 2006;13:169-75.
Isaac-Renton J, Bowie WR, King A, Irwin GS, Ong CS, Fung CP, et al. Detection of Toxoplasma gondii oocysts in drinking water. Appl Environ Microbiol. 1998;64:2278-80. https://doi.org/10.1128/AEM.64.6.2278-2280.1998
Blanco PJ, Assia YM, Montero YM, Orozco KE. ELFA IgG anti-Toxoplasma y PCR anidada para el diagnóstico de toxoplasmosis en mujeres gestantes de Sincelejo, Colombia. Infectio. 2011;15:253-8. https://doi.org/10.1016/S0123-9392(11)70739-1
Benenson MW, Takafuji ET, Lemon SM, Greenup RL, Sulzer AJ. Oocyst-transmitted toxoplasmosis associated with ingestion of contaminated water. N Engl J Med. 1982;307:666-9. https://doi.org/10.1056/NEJM198209093071107
Bowie WR, King AS, Werker DH, Isaac-Renton JL, Bell A, Eng SB, et al. Outbreak of toxoplasmosis associated with municipal drinking water. Lancet. 1997;350:173-7. https://doi.org/10.1016/s0140-6736(96)11105-3
Aramini JJ, Stephen C, Dubey JP, Engelstoft C, Schwantje H, Ribble CS. Potential contamination of drinking water with Toxoplasma gondii oocysts. Epidemiol Infect. 1999;122:305-15. https://doi.org/10.1017/s0950268899002113
De Moura L, García LM, Wada MY, Jones JL, Tuboi SH, Carmo EH, et al. Waterborne, toxoplasmosis, Brazil, from field to gene. Emerg Infect Dis. 2006;12:326-9. https://doi.org/10.3201/eid1202.041115
Balasundaram MB, Andavar R, Palaniswamy M, Venkatapathy N. Outbreak of acquired ocular toxoplasmosis involving 248 patients. Arch. Ophthalmol. 2010;128:28-32. https://doi.org/10.1001/archophthalmol.2009.354
Adamska M. Molecular detection of Toxoplasma gondii in natural surface water bodies in Poland. J Water Health. 2018;16:657-60. https://doi.org/.10.2166/wh.2018.236
Ćirković V, Uzelac A, Miličić D, Klun I, Djurković-Djaković O. First detection of Toxoplasma gondii (Nicolle & Manceaux, 1908) (Eucoccidiorida: Sarcocystidae) in river waters in Serbia. Acta Zool Bulg. 2020;7:79-83. http://www.acta-zoologica-bulgarica.eu/Suppl_15_15
Galvani AT, Guarnieri AP, Padula JA, Funada MR, Silva R, Zanoli MI, et al. Real-time PCR detection of Toxoplasma gondii in surface water samples in São Paulo, Brazil. Parasitol Res. 2019;118:631-40. https://doi.org/10.1007/s00436-018-6185-z
Ministerio del Interior de Colombia. Convenio Interadministrativo N° M-923 de 2013 celebrado entre la nación, ministerio del interior y el Cabildo Mayor Regional del Pueblo Zinú: diagnóstico comunitario y líneas de acción para el plan de salvaguarda étnica del pueblo Zinú, capítulo Córdoba-Sucre y dispersos. Fecha de consulta: 25 de marzo de 2020. Disponible en: https://siic.mininterior.gov.co/sites/default/files/pueblo_zenu_-_diagnostico_comunitario_tomo_1.pdf
Castaño C, González J, Ange C, Zárrate D, Vela M. Ecología y conservación de felinos y presas en el Caribe Colombiano. En: Castaño C, González J, Zárrate D, Ange C, Vela I, editores. Plan de Conservación de Felinos del Caribe colombiano: Los felinos y su papel en la planificación regional integral basada en especies clave. Santa Marta: Fundación Herencia ambiental Caribe, ProCAT Colombia, The Sierra to Sea Institute.; 2013. p. 1-233.
Some similar items:
- Liliana Jazmín Cortés, Sofía Duque, Myriam Consuelo López, Diego Moncada, Diego Molina, Jorge Enrique Gómez-Marín, María Luz Gunturiz, Gene polymorphisms in the dihydrofolate reductase (dhfr) and dihydropteroate synthase (dhps) genes and structural modelling of the dhps gene in Colombian isolates of Toxoplasma gondii , Biomedica: Vol. 34 No. 4 (2014)
- Mercedes Salcedo-Cifuentes, Jesús Cabrera, Yesid Cuesta-Astroz, Edwin Carrascal, Yoshito Eizuru, Martha C. Domínguez, Adalberto Sánchez, Felipe García-Vallejo, Clonal expansion and genomic characterization of the human T-cell lymphotropic virus type I during the integration process in adult T-cell leukemia/lymphoma , Biomedica: Vol. 29 No. 2 (2009)
- Martine Bonnaure-Mallet, Paula Juliana Pérez-Chaparro, Patrice Gracieux, Vincent Meuric, Zohreh Tamanai-Shacoori, Jaime Eduardo Castellanos, Distribution of Porphyromonas gingivalis fimA genotypes in isolates from subgingival plaque and blood sample during bacteremia , Biomedica: Vol. 29 No. 2 (2009)
- Diego Fernando Zea, Martín Prager, Roger Adrian Figueroa, María Consuelo Miranda, Mucosal complication of cutaneous leishmaniasis , Biomedica: Vol. 29 No. 1 (2009)
- Ana Margarita Montalvo, Lianet Monzote, Jorge Fraga, Ivón Montano, Carlos Muskus, Marcel Marín, Simonne De Donck, Iván Darío Vélez, Jean Claude Dujardin, PCR-RFLP and RAPD for typing neotropical Leishmania , Biomedica: Vol. 28 No. 4 (2008)
- Concepción Judith Puerta, Johana María Guevara, Paula Ximena Pavía, Marleny Montilla, Rubén Santiago Nicholls, Edgar Parra, Yuli Katherine Barrera, Evaluation of TcH2AF-R and S35-S36 primers in PCR tests for the detection of Trypanosoma cruzi in mouse cardiac tissue , Biomedica: Vol. 28 No. 4 (2008)
- Marcel Marín, Yudy Alexandra Aguilar, José Robinson Ramírez, Omar Triana, Carlos Enrique Muskus, Molecular and immunological analyses suggest the absence of hydrophilic surface proteins in Leishmania (Viannia) panamensis , Biomedica: Vol. 28 No. 3 (2008)
- Astrid Elena Montoya, José Menco, Natalia Osorio, Maria Alejandra Zuluaga, Juliana Duque, Giovanny Torres, Marcos Restrepo, Concordance between thick blood smear, immunochromatography and polymerase chain reaction for malaria diagnosis , Biomedica: Vol. 28 No. 2 (2008)
- Concepción Judith Puerta, Paula Ximena Pavia, Marleny Montilla, Carolina Flórez, Giomar Herrera, Juan Manuel Ospina, Fred Manrique, Rubén Santiago Nicholls, The first case of congenital Chagas’ disease analyzed by AP-PCR in Colombia , Biomedica: Vol. 29 No. 4 (2009)
- María Teresa Arango, Carlos Jaramillo, María Camila Montealegre, Mabel Helena Bohórquez, María del Pilar Delgado, Genetic characterization of the interleukin 1 β polymorphisms -511, -31 y +3954 in a Colombian population with dyspepsia , Biomedica: Vol. 30 No. 2 (2010)
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |

























